BMG continues to raise awareness of the problem of bias. The following list contains select references and resources relevant to the scope of Bias Methods Group, much of which had been conducted by members of the group. For a more comprehensive list of resources, please consult the Cochrane Library (The Cochrane Methodology Register and the Cochrane Database of Systematic Reviews).
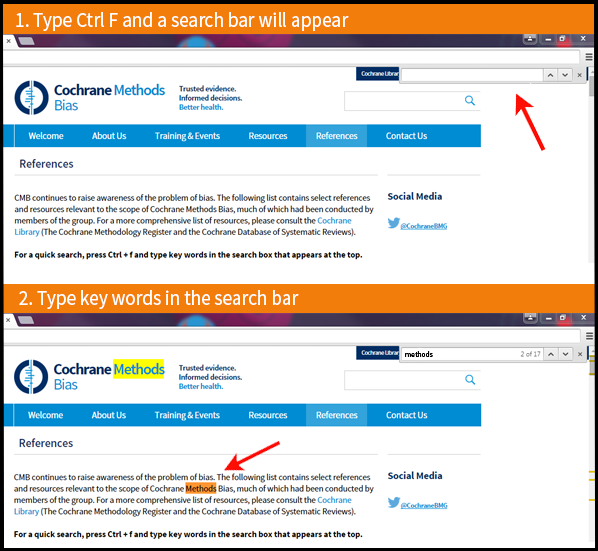
For a quick search of the references:
References are organized by types of bias
Other resources
- Schou IM. Meta-analysis of clinical trials with early stopping: an investigation of potential bias. [Review]. Stat Med 2013 Dec 10;32(28):4859-74. [PMID: 23824994]
- Kimani PK, Todd S, Stallard. Conditionally unbiased estimation in phase II/III clinical trials with early stopping for futility. Stat Med 2013 Jul 30;32(17):2893-910. [PMID: 23413228]
- Hrobjartsson A. Observer bias in randomized clinical trials with time-to-event outcomes: systematic review of trials with both blinded and non-blinded outcome assessors. [Review]. Int J Epidemiol 2014 Jun;43(3):937-48. [PMID: 24448109]
- Murad MH, Montori VM, Ioannidis JP, Jaeschke. How to read a systematic review and meta-analysis and apply the results to patient care: users' guides to the medical literature. JAMA 2014 Jul;312(2):171-9. [PMID: 25005654]
- Wittes J. Stopping a trial early - and then what? Clin Trials 2012 Dec;9(6):714-20. [PMID: 22879573]
- Bafeta A, Dechartres A, Trinquart L, Yavchitz A, Boutron I, Ravaud P. Impact of single centre status on estimates of intervention effects in trials with continuous outcomes: meta-epidemiological study. BMJ 2012;344:e813, 2012.
- Boutron I, Ravaud P. Classification systems to improve assessment of risk of bias. J Clin Epidemiol 2012;65(3):236-8
- Hrobjartsson A,ThomsenAS, Emanuelsson F, Tendal B, Hilden J, Boutron I, et al. Observer bias in randomised clinical trials with binary outcomes: systematic review of trials with both blinded and non-blinded outcome assessors. BMJ 2012;344:e1119, 2012.
- Bascoul-Mollevi C, Laplanche A, Deley MC, Kramar A. SAE: an R package for early stopping rules in clinical trials. Comput Methods Programs Biomed 2011;104(2):243-8.
- Dechartres A, Boutron I, Trinquart L, Charles P, Ravaud P. Single-center trials show larger treatment effects than multicenter trials: evidence from a meta-epidemiologic study. Ann Intern Med 2011;155(1):39-51.
- Hrobjartsson A, Boutron I. Blinding in randomized clinical trials: imposed impartiality. Clin Pharmacol Ther 2011;90(5):732-6
- Bassler D, Briel M, Montori VM, Lane M, Glasziou P, Zhou Q, et al. Stopping randomized trials early for benefit and estimation of treatment effects: systematic review and meta-regression analysis. JAMA 2010;303(12):1180-7
- Berry SM, Carlin BP, Connor J. Bias and trials stopped early for benefit. JAMA 2010;304(2):156-9.
- Boutron I, Dutton S, Ravaud P, Altman DG. Reporting and interpretation of randomized controlled trials with statistically nonsignificant results for primary outcomes. JAMA 2010;303(20):2058-64.
- Ellenberg SS, DeMets DL, Fleming TR. Bias and trials stopped early for benefit. JAMA 2010;304(2):158-9.
- Goodman S, Berry D, Wittes J. Bias and trials stopped early for benefit. JAMA 2010;304(2):157-9.
- Hade EM, Jarjoura D, Lai W. Sample size re-estimation in a breast cancer trial. Clinical Trials 2010;7(3):219-26
- Korn EL, Freidlin B, Mooney M. Bias and trials stopped early for benefit. JAMA 2010;304(2):157-8.
- Schroen AT, Petroni GR, Wang H, Gray R, Wang XF, Cronin W, et al. Preliminary evaluation of factors associated with premature trial closure and feasibility of accrual benchmarks in phase III oncology trials. Clinical Trials 2010;7(4):312-21.
- Atkins CD. Stopping trials early for positive results: the need to know how much. J Clin Oncol 2009;27(21):e29.
- Briel M, Lane M, Montori VM, Bassler D, Glasziou P, Malaga G, et al. Stopping randomized trials early for benefit: a protocol of the Study Of Trial Policy Of Interim Truncation-2 (STOPIT-2). Trials 2009;10:49, 2009.
- Ergina PL, Cook JA, Blazeby JM, Boutron I, Clavien PA, Reeves BC, et al. Challenges in evaluating surgical innovation. Lancet 2009;374(9695):1097-104.
- Fernandes RM, Lee JH, Offringa M. A systematic review of the reporting of Data Monitoring Committees' roles, interim analysis and early termination in pediatric clinical trials. BMC Pediatr 2009;9:77
- Freidlin B, Korn EL. Stopping clinical trials early for benefit: impact on estimation. Clinical Trials 2009;6(2):119-25.
- Hirsh J, Guyatt G. Clinical experts or methodologists to write clinical guidelines? Lancet 2009;374(9686):273-5.
- Korn EL, Freidlin B, Mooney M. Stopping or reporting early for positive results in randomized clinical trials: The National Cancer Institute Cooperative Group experience from 1990 to 2005. J Clin Oncol 2009;27(10):1712-21.
- Kramar A, Bascoul-Mollevi C. Early stopping rules in clinical trials based on sequential monitoring of serious adverse events. Med Decis Making 2009;29(3):343-50.
- Bassler D, Montori VM, Briel M, Glasziou P, Guyatt G. Early stopping of randomized clinical trials for overt efficacy is problematic. J Clin Epidemiol 2008;61(3):241-6.
- Fleming TR, Sharples K, McCall J, Moore A, Rodgers A, Stewart R. Maintaining confidentiality of interim data to enhance trial integrity and credibility. Clinical Trials 2008;5(2):157-67.
- Floriani I, Rotmensz N, Albertazzi E, Torri V, Rosa M, Tomino C, et al. Approaches to interim analysis of cancer randomised clinical trials with time to event endpoints: a survey from the Italian National Monitoring Centre for Clinical Trials. Trials 2008
- Goodman SN. Systematic reviews are not biased by results from trials stopped early for benefit. J Clin Epidemiol 2008;61(1):95-6.
- Schunemann HJ, Kirpalani H, Rozenberg A, Guyatt GH. Systematic reviewers neglect bias that results from trials stopped early for benefit. Journal of Clinical Epidemiology 2007; 60(9):869-73.
- Shepherd R, Macer JL, Grady D. Planning for closeout - from Day One. Contemporary Clinical
- Taylor HA, Chaisson L, Sugarman J. Enhancing communication among data monitoring committees and institutional review boards. Clinical Trials 2008;5(3):277-82.
- Tharmanathan P, Calvert M, Hampton J, Freemantle N. The use of interim data and Data Monitoring Committee recommendations in randomized controlled trial reports: frequency, implications and potential sources of bias. BMC Medical Research Methodology 2008;8:12.
- Trotta F, Apolone G, Garattini S, Tafuri G. Stopping a trial early in oncology: for patients or for industry? Ann Oncol 2008;19(7):1347-53.
- Catala-Lopez F. When are statins cost-effective in cardiovascular prevention? A systematic review of sponsorship bias and conclusions in economic evaluations of statins. [Review]. PLoS ONE 2013;8(7):e69462. [PMID: 23861972]
- Macleod M. Some salt with your statin, professor? PLoS Biol 2014 Jan;12(1):e1001768. [PMID: 24465176]
- Krauth D. Nonindustry-sponsored preclinical studies on statins yield greater efficacy estimates than industry-sponsored studies: a meta-analysis. PLoS Biol 2014 Jan;12(1):e1001770. [PMID: 24465178]
- Naci H. Industry sponsorship bias in research findings: a network meta-analysis of LDL cholesterol reduction in randomised trials of statins. BMJ 2014;349:g5741. [PMID: 25281681]
- Lundh A, Krogsboll LT, Gotzsche PC. Sponsors' participation in conduct and reporting of industry trials: a descriptive study. Trials 2012;13:146. [PMID: 22920226]
- Bindslev JB, Schroll J, Gotzsche PC, Lundh A. Underreporting of conflicts of interest in clinical practice guidelines: cross sectional study. BMC Med Ethics 2013;14:19. [PMID: 23642105]
- Lundh A, Hrobjartsson A, Gotzsche PC. Income from reprints creates a conflict of interests. BMJ 2012;345:e4970, 2012.
- Peura PK, Martikainen JA, Purmonen TT, Turunen JH. Sponsorship-related outcome selection bias in published economic studies of triptans: systematic review. Med Decis Making 2012;32(2):237-45.
- Aldairy Y,Nguyen PL, Jatoi A. Bone pain from granulocyte colony stimulating factor: does clinical trial sponsorship by a pharmaceutical company influence its reporting? Eur J Cancer Care (Engl ) 2011;20(1):72-6.
- Becker A, Dorter F, Eckhardt K, Viniol A, Baum E, Kochen MM, et al. The association between a journal's source of revenue and the drug recommendations made in the articles it publishes. CMAJ 2011;183(5):544-8.
- Jinapriya D, Anraku A, Alasbali T, Trope GE, Buys YM. Evaluation of investigator bias in industry-funded clinical trials of latanoprost. Can J Ophthalmol 2011;46(6):531-6.
- Lunde IM, Gotzsche PC. Transparency, reliability and conflict of interest. Ugeskr Laeger 2011;173(33):1989, 2011.
- Lundh A, Krogsboll LT, Gotzsche PC. Access to data in industry-sponsored trials. Lancet 2011;378(9808):1995-6.
- Rodriguez JM, Paz S, Lizan L, Gonzalez P. The use of quality-adjusted life-years in the economic evaluation of health technologies inSpain: a review of the 1990-2009 literature. Value Health 2011;14(4):458-64.
- Roseman M, Milette K, Bero LA, Coyne JC, Lexchin J, Turner EH, et al. Reporting of conflicts of interest in meta-analyses of trials of pharmacological treatments. JAMA 2011;305(10):1008-17.
- Toal MJ. Industry sponsored bias: NICE may be biased too. BMJ 2011;342:d474, 2011.
- Als-Nielsen B, Chen W, Gluud C, Kjaergard LL. Association of funding and conclusions in randomised drug trials. A reflection of treatment effect or adverse events? JAMA 2003; 290: 921-28.
- Bero L, Oostvogel F, Bacchetti P, Lee K (2007) Factors associated with findings of published trials of drug?drug comparisons: Why some statins appear more efficacious than others. PLoS Med 4(6): e184. doi:10.1371/journal.pmed.0040184 .
- Bhandari M, Busse JW, Jackowski D, Montori VM, Schunemann H, Sprague S, et al. Association between industry funding and statistically significant pro-industry findings in medical and surgical randomized trials. CMAJ Canadian Medical Association Journal 2004; 170(4): 477-80.
- Cho MK, Bero LA. The quality of drug studies published in symposium proceedings. Ann Intern Med 1996; 124(5): 485-9.
- Davidson RA. Source of funding and outcome of clinical trials. Journal of General Internal Medicine 1986; 1(3): 155-8.
- DelMC. Commentary: But what should journals actually do to keep industry sponsored research unbiased? BMJ 2010;341:c5406, 2010
- Dias S,WeltonNJ, Ades AE. Study designs to detect sponsorship and other biases in systematic reviews. J Clin Epidemiol 2010;63(6):587-8
- Djulbegovic B, Lacevic M, Cantor A, Fields KK, Bennett CL, Adams JR, Kuderer NM, Lyman GH (2000). The uncertainty principle and industry-sponsored research. Lancet 356:635-638.
- Gartlehner G, Morgan L, Thieda P, Fleg A. The effect of study sponsorship on a systematically evaluated body of evidence of head-to-head trials was modest: secondary analysis of a systematic review. J Clin Epidemiol 2010;63(2):117-25.
- Gartlehner G, Fleg A. Comparative effectiveness reviews and the impact of funding bias. J Clin Epidemiol 2010;63(6):589-90.
- Hemminki E (1980). Study of information submitted by drug companies to licensing authorities. BMJ 280:833-6.
- John-Baptiste A, Bell C. Industry sponsored bias in cost effectiveness analyses. BMJ 2010;341:c5350, 2010.
- Krzyzanowska MK, Pintilie M, Tannock IF. Factors associated with failure to publish large randomized trials presented at an oncology meeting. JAMA 2003; 290(4): 495-501.
- Lexchin J, Bero LA, Djulbegovic B, Clark O. Pharmaceutical industry sponsorship and research outcome and quality: systematic review. BMJ 2003; 326(7400 ): 1167-70.
- Lesser LI. Reducing potential bias in industry-funded nutrition research. Am J Clin Nutr 2009;90(3):699-700.
- Liu XM, Li YP,Wu TX, Liu GJ, Li J. A Survey of the Status of Funding of Registered Chinese Clinical Trials .Chinese Journal of Evidence-Based Medicine, 2008; 8(5):305-311.
- Lundh A, Barbateskovic M, Hrobjartsson A, Gotzsche PC. Conflicts of interest at medical journals: the influence of industry-supported randomised trials on journal impact factors and revenue - cohort study. PLoS Med 2010;7(10):e1000354, 2010.
- McPartland JM. Obesity, the endocannabinoid system, and bias arising from pharmaceutical sponsorship. PLoS One 2009;4(3):e5092, 2009. Available
- Melander H, hlqvist-Rastad J, Meijer G, Beermann B. Evidence b(i)ased medicine--selective reporting from studies sponsored by pharmaceutical industry: review of studies in new drug applications. BMJ 2003; 326(7400 ): 1171-3.
- Vedula SS, Bero L, Scherer RW, Dickersin K. Outcome reporting in industry-sponsored trials of gabapentin for off-label use. N Engl J Med 2009;361(20):1963-71
- Angell M. Industry-sponsored clinical research: a broken system. JAMA 2008;300(9):1069-71.
- Berger E. Ghostwriters, data manipulation and dollar diplomacy: how drug companies pull the strings in clinical research. Ann Emerg Med 2008;52(2):137-9.
- DavisJM, Chen N, Glick ID. Issues that may determine the outcome of antipsychotic trials: industry sponsorship and extrapyramidal side effect. Neuropsychopharmacology 2008;33(5):971-5.
- Doucet M, Sismondo S. Evaluating solutions to sponsorship bias. J Med Ethics 2008;34(8):627-30.
- Hammerschmidt D. Bias in the design, interpretation, and publication of industry-sponsored clinical research.MinnMed 2008;91(6):46-7.
- Jorgensen AW, Maric KL, Tendal B, Faurschou A, Gotzsche PC. Industry-supported meta-analyses compared with meta-analyses with non-profit or no support: differences in methodological quality and conclusions. BMC Med Res Methodol 2008;8:60, 2008.
- Kaufman SR. Bias and sponsored research. Ophthalmology 2008;115(2):412-3.
- Rowbotham MC. The impact of selective publication on clinical research in pain. Pain 2008;140(3):401-4.
- Saito M, Hasegawa R. Survey of the amount of the industry funding for biomedical research - analysis of questionnaire for medical and pharmaceutical departments, professors belonging these departments and the pharmaceutical companies. Bulletin of National
- Taira BR, Jahnes K, Singer AJ, McLarty AJ. Does reported funding differ by gender in the surgical literature? Ann Surg 2008;247(6):1069-73.
- Thomas O, Thabane L, Douketis J,ChuR, Westfall AO, Allison DB. Industry funding and the reporting quality of large long-term weight loss trials. International Journal of Obesity 2008;32(10):1531-6.
- Perlis RH, Perlis CS, Wu Y, Hwang C, Joseph M, Nierenberg AA. Industry sponsorship and financial conflict of interest in the reporting of clinical trials in psychiatry. American Journal of Psychiatry 2005; 162(10): 1957-60.
- Rochon PA, Gurwitz JH, Simms RW, Fortin PR, Felson DT, Minaker KL, et al. A study of manufacturer-supported trials of nonsteroidal anti-inflammatory drugs in the treatment of arthritis. Arch Intern Med 1994; 154(2): 157-63.
- Wolf, FM, Lynch JR; Cunningham MRA; Warme WJ; Schaad DC, Leopold SS. Commercially-funded and United States-based research is more likely to be published; good-quality studies with negative outcomes are not. J Bone Joint Surg; 2007:89-A(5): 110-18.
- Wolf FM, Cunningham MR, Warme,WJ, Schaad DC, Leopold S.S. Industry-funded positive studies not associated with better design or larger size. Clin Orthop Relat Res 2007; 457:235-41.
- Tsilidis KK, Panagiotou OA, Sena ES, Aretouli E, Evangelou. Evaluation of excess significance bias in animal studies of neurological diseases. PLoS Biol 2013 Jul;11(7):e1001609. [PMID: 23874156]
- Forbes D. Blinding: an essential component in decreasing risk of bias in experimental designs. Evid Based Nurs 2013 Jul;16(3):70-1. [PMID: 23696228]
- Zhao W. Selection bias, allocation concealment and randomization design in clinical trials. Contemp Clin Trials 2013 Sep;36(1):263-5. [PMID: 23871796]
- Jordan S, Watkins. Volunteer bias in recruitment, retention, and blood sample donation in a randomised controlled trial involving mothers and their children at six months and two years: a longitudinal analysis. PLoS ONE 2013;8(7):e67912. [PMID: 23874465]
- Lockyer S, Hodgson. "Spin" in wound care research: the reporting and interpretation of randomized controlled trials with statistically non-significant primary outcome results or unspecified primary outcomes. Trials 2013;14:371. [PMID: 24195770]
- Pressman MR. Sleepwalking, amnesia, comorbid conditions and triggers: effects of recall and other methodological biases. Sleep 2013 Nov;36(11):1757-8. [PMID: 24179310]
- Jacobs WC, Kruyt MC, Moojen WA, Verbout AJ, Oner FC. No evidence for intervention-dependent influence of methodological features on treatment effect. [Review]. J Clin Epidemiol 2013 Dec;66(12):1347-55. [PMID: 24018348]
- Oomens MA, Heymans MW, Forouzanfar. Risk of bias in research in oral and maxillofacial surgery. [Review]. Br J Oral Maxillofac Surg 2013 Dec;51(8):913-9. [PMID: 23746677]
- Takabayashi N. Biased safety reporting in blinded randomized clinical trials: meta-analysis of angiotensin receptor blocker trials. PLoS ONE 2013;8(9):e75027. [PMID: 24086427]
- Krauth D. Instruments for assessing risk of bias and other methodological criteria of published animal studies: a systematic review. [Review]. Environ Health Perspect 2013 Sep;121(9):985-92. [PMID: 23771496]
- Hartling L. Testing the risk of bias tool showed low reliability between individual reviewers and across consensus assessments of reviewer pairs. J Clin Epidemiol 2013 Sep;66(9):973-81. [PMID: 22981249]
- Chaimani A. Effects of study precision and risk of bias in networks of interventions: a network meta-epidemiological study. [Review]. Int J Epidemiol 2013 Aug;42(4):1120-31. [PMID: 23811232]
- Zeisser C. Methodological biases in estimating the relationship between alcohol consumption and breast cancer: the role of drinker misclassification errors in meta-analytic results. Alcohol Clin Exp Res 2014 Aug;38(8):2297-306. [PMID: 25156617]
- Ioannidis JP, Greenland S, Hlatky MA, Khoury MJ, Macleod MR, Moher. Increasing value and reducing waste in research design, conduct, and analysis. Lancet 2014 Jan 11;383(9912):166-75. [PMID: 24411645]
- Favalli EG. The comparison of effects of biologic agents on rheumatoid arthritis damage progression is biased by period of enrollment: data from a systematic review and meta-analysis. [Review]. Semin Arthritis Rheum 2014 Jun;43(6):730-7. [PMID: 24332807]
- Armijo-Olivo S, Cummings GG, Fuentes. Identifying items to assess methodological quality in physical therapy trials: a factor analysis. Phys Ther 2014 Sep;94(9):1272-84. [PMID: 24786942]
- Ochodo EA, van Enst WA, Naaktgeboren CA, de Groot JA, Hooft. Incorporating quality assessments of primary studies in the conclusions of diagnostic accuracy reviews: a cross-sectional study. BMC Med Res Methodol 2014;14:33. [PMID: 24588874]
- Avni T. Assessment of bias in outcomes reported in trials on pneumonia: a systematic review. [Review]. Eur J Clin Microbiol Infect Dis 2014 Jun;33(6):969-74. [PMID: 24352842]
- Savovic J. Evaluation of the Cochrane Collaboration's tool for assessing the risk of bias in randomized trials: focus groups, online survey, proposed recommendations and their implementation. Syst 2014;3:37. [PMID: 24731537]
- Margulis AV. Quality assessment of observational studies in a drug-safety systematic review, comparison of two tools: the Newcastle-Ottawa Scale and the RTI item bank. Clin Epidemiol 2014;6:359-68. [PMID: 25336990]
- Sinha YK, Craig JC, Sureshkumar. Risk of bias in randomized trials of pharmacological interventions in children and adults. J Pediatr 2014 Aug;165(2):367-71. [PMID: 24814415]
- Clark L. A methodological review of recent meta-analyses has found significant heterogeneity in age between randomized groups. J Clin Epidemiol 2014 Sep;67(9):1016-24. [PMID: 24909873]
- Glasziou P. Reducing waste from incomplete or unusable reports of biomedical research. Lancet 2014 Jan 18;383(9913):267-76. [PMID: 24411647]
- Koretz RL. The presence and effect of bias in trials of early enteral nutrition in critical care. [Review]. Clin Nutr 2014 Apr;33(2):240-5. [PMID: 23845382]
- Bello S, Krogsboll LT, Gruber. Lack of blinding of outcome assessors in animal model experiments implies risk of observer bias. [Review]. J Clin Epidemiol 2014 Sep;67(9):973-83. [PMID: 24972762]
- Mathieu E, Herbert RD, McGeechan. A theoretical analysis showed that blinding cannot eliminate potential for bias associated with beliefs about allocation in randomized clinical trials. J Clin Epidemiol 2014 Jun;67(6):667-71. [PMID: 24767518]
- Hartling L. Quantifying bias in randomized controlled trials in child health: a meta-epidemiological study. PLoS ONE 2014;9(2):e88008. [PMID: 24505351]
- Hopewell S, Boutron I, Altman DG, Ravaud P. Incorporation of assessments of risk of bias of primary studies in systematic reviews of randomised trials: a cross-sectional study. BMJ Open 2013;3(8):e003342. [PMID: 23975265]
- Hu D, Kang DY, Wu YX. [Methodological quality assessment of systematic reviews correlated to traditional Chinese medicine published in China]. [Chinese]. Zhongguo Zhong Xi Yi Jie He Za Zhi 2011 Mar;31(3):402-6. [PMID: 21485088]
- Seo HJ, Kim KU. Quality assessment of systematic reviews or meta-analyses of nursing interventions conducted by Korean reviewers. BMC Med Res Methodol 2012;12:129. [PMID: 22928687]
- Thorlund K, Imberger G, Johnston BC, Walsh M, Awad T, Thabane L, et al. Evolution of heterogeneity (I2) estimates and their 95% confidence intervals in large meta-analyses. PLoS ONE 2012;7(7):e39471. [PMID: 22848355]
- Glujovsky D, Riestra B, Coscia A, Boggino C, Comande D, Ciapponi A. Assessment of research quality inmajor infertility journals. Fertil Steril 2012 Dec;98(6):1539-43. [PMID: 22981173]
- Hrobjartsson A, Boutron I, Turner L, Altman DG, Moher D. Assessing risk of bias in randomised clinical trials included in Cochrane Reviews: The why is easy, the how is a challenge. Cochrane Database Syst Rev 2013;4:ED000058. [PMID: 23728703]
- Chung JH, Lee SW. Assessing the quality of randomized controlled urological trials conducted by korean medical institutions. Korean J Urol 2013 May;54(5):289-96. [PMID: 23700493]
- Chess LE, Gagnier J. Risk of bias of randomized controlled trials published in orthopaedic journals. BMC Med Res Methodol 2013;13:76. [PMID: 23758875]
- Woods S, Coghlan A, Rivers D, Warnecke T, Jeffries SJ, Kwon T, et al. Duplication and retention biases of essential and non-essential genes revealed by systematic knockdown analyses. PLoS Genet 2013 May;9(5):e1003330. [PMID: 23675306]
- Moller AM. [Risk of bias by blinded vs unblinded evaluation of articles in systematic reviews]. [Danish]. Ugeskr Laeger 2013 Apr 1;175(14):951-4. [PMID: 23582068]
- Vale CL, Tierney JF, Burdett S. Can trial quality be reliably assessed from published reports of cancer trials: evaluation of risk of bias assessments in systematic reviews. BMJ 2013;346:f1798. [PMID: 23610376]
- Kim SY, Park JE, Lee YJ, Seo HJ, Sheen SS, Hahn S, et al. Testing a tool for assessing the risk of bias for nonrandomized studies showed moderate reliability and promising validity. J Clin Epidemiol 2013 Apr;66(4):408-14. [PMID: 23337781]
- Hrobjartsson A, Thomsen AS, Emanuelsson F, Tendal B, Hilden J, Boutron I, et al. Observer bias in randomized clinical trials with measurement scale outcomes: a systematic review of trials with both blinded and nonblinded assessors. [Review]. CMAJ 2013 Mar 5;185(4):E201-E211. [PMID: 23359047]
- Dechartres A, Trinquart L, Boutron I, Ravaud P. Influence of trial sample size on treatment effect estimates: meta-epidemiological study. [Review]. BMJ 2013;346:f2304. [PMID: 23616031]
- Shepherd BE, Shaw PA, Dodd LE. Using audit information to adjust parameter estimates for data errors in clinical trials. Clin Trials 2012 Dec;9(6):721-9. [PMID: 22848072]
- Savovic J, Jones H, Altman D, Harris R, Juni P, Pildal J, et al. Influence of reported study design characteristics on intervention effect estimates from randomised controlled trials: combined analysis of meta-epidemiological studies. [Review]. Health Technol Assess 2012 Sep;16(35):1-82. [PMID: 22989478]
- Hrobjartsson A, Thomsen AS, Emanuelsson F, Tendal B, Hilden J, Boutron I, et al. Observer bias in randomized clinical trials with measurement scale outcomes: a systematic review of trials with both blinded and nonblinded assessors. CMAJ 2013;185(4):E201-E211.
- Panagiotou OA, Contopoulos-Ioannidis DG, & Ioannidis J. Comparative effect sizes in randomised trials from less developed and more developed countries: meta-epidemiological assessment. BMJ: British Medical Journal 2013; 346.
- Schoorl M, Putman P, Van Der Does W. Attentional bias modification in posttraumatic stress disorder: a randomized controlled trial. Psychother Psychosom 2013;82(2):99-105.
- Adams S, Ataya AF,AttwoodAS, Munafo MR. Effects of acute alcohol consumption on alcohol-related cognitive biases in light and heavy drinkers are task-dependent. J Psychopharmacol 2012;26(2):245-53
- Carlbring P, Apelstrand M, Sehlin H, Amir N, Rousseau A, Hofmann SG, et al. Internet-delivered attention bias modification training in individuals with social anxiety disorder--a double blind randomized controlled trial. BMC Psychiatry 2012;12:66, 2012
- Carrasco M, Volkmar FR, Bloch MH. Pharmacologic treatment of repetitive behaviors in autism spectrum disorders: evidence of publication bias. Pediatrics 2012;129(5):e1301-e1310
- De Oliveira GSJ, Chang R,KendallMC, Fitzgerald PC, McCarthy RJ. Publication bias in the anesthesiology literature. Anesth Analg 2012;114(5):1042-8.
- Dicus JR, Seegmiller JG. Unanticipated ankle inversions are significantly different from anticipated ankle inversions during drop landings: overcoming anticipation bias. J 2012;APPL. BIOMECH.. 28(2):148-55.
- Fernie G, Christiansen P, Cole JC, Rose AK, Field M. Effects of 0.4 g/kg alcohol on attentional bias and alcohol-seeking behaviour in heavy and moderate social drinkers. J Psychopharmacol 2012;26(7):1017-25.
- Kamboj SK, Joye A, Das RK, Gibson AJ, Morgan CJ, Curran HV. Cue exposure and response prevention with heavy smokers: a laboratory-based randomised placebo-controlled trial examining the effects of D-cycloserine on cue reactivity and attentional bias. Psychopharmacology (Berl ) 2012;221(2):273-84.
- Kometer M, Schmidt A, Bachmann R, Studerus E, Seifritz E, Vollenweider FX. Psilocybin biases facial recognition, goal-directed behavior, and mood state toward positive relative to negative emotions through different serotonergic subreceptors. Biol Psychiatry 2012;72(11):898-906.
- Luijten M, Veltman DJ, Hester R, Smits M, Pepplinkhuizen L, Franken IH. Brain activation associated with attentional bias in smokers is modulated by a dopamine antagonist. Neuropsychopharmacology 2012;37(13):2772-9
- McGivern RF, Adams B, Handa RJ, Pineda JA. Men and women exhibit a differential bias for processing movement versus objects. PLoS One 2012;7(3):e32238, 2012.
- Meerpohl JJ, Langer G, Perleth M, Gartlehner G, Kaminski-Hartenthaler A, Schunemann H. GRADE guidelines: 4. Rating the quality of evidence - limitations of clinical trials (risk of bias). Z 2012;Evid. Fortbild. Qual. Gesundh.wes.. 106(6):457-69.
- Moss-RacusinCA, Dovidio JF, Brescoll VL, Graham MJ, Handelsman J. Science faculty's subtle gender biases favor male students. Proc Natl Acad Sci U S A 2012;109(41):16474-9.
- Nathan PJ, O'Neill BV, Mogg K, Bradley BP, Beaver J, Bani M, et al. The effects of the dopamine D3 receptor antagonist GSK598809 on attentional bias to palatable food cues in overweight and obese subjects. Int J Neuropsychopharmcol 2012;15(2):149-61.
- Pandis N. Randomization. Part 3: allocation concealment and randomization implementation. Am J Orthod Dentofacial Orthop 2012;141(1):126-8.
- Schmidt HG, Muijtjens AM, Van der Vleuten CP, Norman GR. Differential student attrition and differential exposure mask effects of problem-based learning in curriculum comparison studies. Acad Med 2012;87(4):463-75.
- Skougaard K, McCullagh MJ, Nielsen D, Hendel HW, Jensen BV, Johannesen HH. Observer variability in a phase II trial - assessing consistency in RECIST application. Acta Oncol 2012;51(6):774-80.
- Tamm M, Cramer E,Kennes LN, Heussen N. Influence of selection bias on the test decision. A simulation study. Methods Inf Med 2012;51(2):138-43.
- Terbeck S, Kahane G, McTavish S, Savulescu J, Cowen PJ, Hewstone M. Propranolol reduces implicit negative racial bias. Psychopharmacology (Berl ) 2012;222(3):419-24.
- van Rees LJ, Ballard KJ, McCabe P, Macdonald-D'Silva AG, Arciuli J. Training production of lexical stress in typically developing children using orthographically biased stimuli and principles of motor learning. Am J Speech Lang Pathol 2012;21(3):197-206.
- Alon S, Vigneault F, Eminaga S,ChristodoulouDC, Seidman JG, Church GM, et al. Barcoding bias in high-throughput multiplex sequencing of miRNA. Genome Res 2011;21(9):1506-11
- Beard C, Weisberg RB, Amir N. Combined cognitive bias modification treatment for social anxiety disorder: a pilot trial. Depress Anxiety 2011;28(11):981-8.
- Ferreira CA, Loureiro CA, Saconato H, Atallah A. Validity of Qualis database as a predictor of evidence hierarchy and risk of bias in randomized controlled trials: a case study in dentistry. Clinics 2011;66(2):337-42
- Ferreira CA, Loureiro CA, Saconato H, Atallah AN. Assessing the risk of bias in randomized controlled trials in the field of dentistry indexed in the Lilacs (Literatura Latino-Americana e do Caribe em Ciencias da Saude) database.Sao PauloMed J 2011;129(
- Guyatt GH, Oxman AD, Vist G, Kunz R, Brozek J, Alonso-Coello P, et al. GRADE guidelines: 4. Rating the quality of evidence--study limitations (risk of bias). J Clin Epidemiol 2011;64(4):407-15.
- Hartling L, Bond K, Vandermeer B, Seida J, Dryden DM, Rowe BH. Applying the risk of bias tool in a systematic review of combination long-acting beta-agonists and inhaled corticosteroids for persistent asthma. PLoS One 2011;6(2):e17242, 2011.
- Herbison P, Hay-Smith J, Gillespie WJ. Different methods of allocation to groups in randomized trials are associated with different levels of bias. A meta-epidemiological study. J Clin Epidemiol 2011;64(10):1070-5.
- Higgins JP, Altman DG, Gotzsche PC, Juni P, Moher D, Oxman AD, et al. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ 2011;343:d5928, 2011
- Jinapriya D, Anraku A, Alasbali T, Trope GE, Buys YM. Evaluation of investigator bias in industry-funded clinical trials of latanoprost. Can J Ophthalmol 2011;46(6):531-6.
- Kamboj SK, Massey-Chase R, Rodney L, Das R, Almahdi B, Curran HV, et al. Changes in cue reactivity and attentional bias following experimental cue exposure and response prevention: a laboratory study of the effects of D-cycloserine in heavy drinkers. Psychopharmacology (Berl ) 2011;217(1):25-37.
- Kinoshita S, Norris D. Does the familiarity bias hypothesis explain why there is no masked priming for "NO" decisions? Mem Cognit 2011;39(2):319-34.
- Lambert J. Statistics in brief: how to assess bias in clinical studies? Clin Orthop Relat Res 2011;469(6):1794-6.
- Liu CJ, LaValley M, Latham NK. Do unblinded assessors bias muscle strength outcomes in randomized controlled trials of progressive resistance strength training in older adults? Am J Phys Med Rehabil 2011;90(3):190-6
- Liu Y, Yang S, Dai J, Xu Y, Zhang R, Jiang H, et al. Risk of bias tool in systematic reviews/meta-analyses of acupuncture in Chinese journals. PLoS One 2011;6(12):e28130
- Mahoney CR, Brunye TT, Giles G, Lieberman HR, Taylor HA. Caffeine-induced physiological arousal accentuates global processing biases. Pharmacol Biochem Behav 2011;99(1):59-65.
- Odgaard-Jensen J, Vist GE, Timmer A, Kunz R, Akl EA, Schunemann H, et al. Randomisation to protect against selection bias in healthcare trials. Cochrane Database Syst Rev 2011;(4):MR000012, 2011
- Robertson CT. The money blind: how to stop industry bias in biomedical science, without violating the First Amendment. Am J Law Med 2011;37(2-3):358-87.
- Song JW, Kim HM, Bellfi LT, Chung KC. The effect of study design biases on the diagnostic accuracy of magnetic resonance imaging for detecting silicone breast implant ruptures: a meta-analysis. Plast Reconstr Surg 2011;127(3):1029-44
- Taves DR. Minimization does not by its nature preclude allocation concealment and invite selection bias, as Berger claims. Contemp Clin Trials 2011;32(3):323, 2011.
- Thompson WK, Holland D, Alzheimer's Disease Neuroimaging Initiative. Bias in tensor based morphometry Stat-ROI measures may result in unrealistic power estimates. Neuroimage 2011;57(1):1-4.
- Vaux DL. A biased comment on double-blind review. Br J Dermatol 2011;165(3):454, 2011.
- Yu X, Ivanic J, Memisevic V, Wallqvist A, Reifman J. Categorizing biases in high-confidence high-throughput protein-protein interaction data sets. Mol Cell Proteomics 2011;10(12):M111.
- Anker CJ, Hymas RV, Hazard LJ, Boucher KM, Jensen RL, Shrieve DC. Stereotactic radiosurgery eligibility and selection bias in the treatment of glioblastoma multiforme. J Neurooncol 2010;98(2):253-63.
- Berger VW. Randomization, permuted blocks, masking, allocation concealment, and selection bias in the Tobacco Quit Line Study. Contemp Clin Trials 2010;31(3):201
- Berger VW. Minimization, by its nature, precludes allocation concealment, and invites selection bias. Contemp Clin Trials 2010;31(5):406.
- Chaouachi K. Hookah (shisha, narghile, "water pipe") indoor air contamination in German unrealistic experiment. Serious methodological biases and ethical concern. Food Chem Toxicol 2010;48(3):992-5
- Cossio P, Marinelli F, Laio A, Pietrucci F. Optimizing the performance of bias-exchange metadynamics: folding a 48-residue LysM domain using a coarse-grained model. J 2010;phys. chem., B Condens. mater. surf. interfaces biophys.. 114(9):3259-65.
- Crocetti MT, Amin DD, Scherer R. Assessment of risk of bias among pediatric randomized controlled trials. Pediatrics 2010;126(2):298-305
- Ersche KD, Bullmore ET, Craig KJ, Shabbir SS, Abbott S, Muller U, et al. Influence of compulsivity of drug abuse on dopaminergic modulation of attentional bias in stimulant dependence. Arch Gen Psychiatry 2010;67(6):632-44.
- Fernandes LP, Annibale A, Kleinjung J, Coolen AC, Fraternali F. Protein networks reveal detection bias and species consistency when analysed by information-theoretic methods. PLoS One 2010;5(8):e12083, 2010
- Hester R, Lee N, Pennay A, Nielsen S, Ferris J. The effects of modafinil treatment on neuropsychological and attentional bias performance during 7-day inpatient withdrawal from methamphetamine dependence. Exp Clin Psychopharmacol 2010;18(6):489-97.
- Hsu VM, Khanna D, Smith E, Filemon T, Whelton S, Lopata M, et al. Use of a medication control officer to reduce bias in a clinical trial: lessons learned from the scleroderma lung study. Clin trials2010; 7(1):85-9.
- Lundh A, Barbateskovic M, Hrobjartsson A, Gotzsche PC. Conflicts of interest at medical journals: the influence of industry-supported randomised trials on journal impact factors and revenue - cohort study. PLoS Med 2010;7(10):e1000354, 2010.
- Menzies-Gow NJ, Stevens KB, Sepulveda MF, Jarvis N, Marr CM. Repeatability and reproducibility of the Obel grading system for equine laminitis. Vet Rec 2010;167(2):52-5.
- Montgomery C, Field M, Atkinson AM, Cole JC, Goudie AJ, Sumnall HR. Effects of alcohol preload on attentional bias towards cocaine-related cues. Psychopharmacology (Berl ) 2010;210(3):365-75.
- Psaty BM, Prentice RL. Minimizing bias in randomized trials: the importance of blinding. JAMA 2010;304(7):793-4
- Suri P, Hunter DJ, Katz JN, Li L, Rainville J. Bias in the physical examination of patients with lumbar radiculopathy. BMC Musculoskelet Disord 2010;11:275, 2010
- Tedersoo L, Nilsson RH, Abarenkov K, Jairus T, Sadam A, Saar I, et al. 454 Pyrosequencing and Sanger sequencing of tropical mycorrhizal fungi provide similar results but reveal substantial methodological biases. New phytol 2010;188(1):291-301
- van der Aa MN, Steyerberg EW, Bangma C, van Rhijn BW, Zwarthoff EC, van der Kwast TH. Cystoscopy revisited as the gold standard for detecting bladder cancer recurrence: diagnostic review bias in the randomized, prospective CEFUB trial. J Urol 2010;183(1):
- van Peer JM, Spinhoven P, Roelofs K. Psychophysiological evidence for cortisol-induced reduction in early bias for implicit social threat in social phobia. Psychoneuroendocrinology 2010;35(1):21-32.
- Yang SS, Valdes-Lopez O, Xu WW, Bucciarelli B, Gronwald JW, Hernandez G, et al. Transcript profiling of common bean (Phaseolus vulgaris L.) using the GeneChip Soybean Genome Array: optimizing analysis by masking biased probes. BMC plant biol 2010;10:85.
- Yang SS, Xu WW, Tesfaye M, Lamb JF, Jung HJ, VandenBosch KA, et al. Transcript profiling of two alfalfa genotypes with contrasting cell wall composition in stems using a cross-species platform: optimizing analysis by masking biased probes. BMC Genomics 20
- Blackwell SC, Thompson L, Refuerzo J. Full publication of clinical trials presented at a national maternal-fetal medicine meeting: is there a publication bias? Am J Perinatol 2009;26(9):679-82.
- Boehmer J, Yong P. How well does blinding work in randomized controlled trials?: a counterpoint. Clin Pharmacol Ther 2009;85(5):463-5.
- Campbell-Yeo M, Ranger M, Johnston C, Fergusson D. Controlling bias in complex nursing intervention studies: a checklist. Can J Nurs Res 2009;41(4):32-50.
- Dickinson CA, Intraub H. Spatial asymmetries in viewing and remembering scenes: consequences of an attentional bias? Atten Percept Psychophys 2009;71(6):1251-62.
- Firk C, Markus CR. Differential effects of 5-HTTLPR genotypes on mood, memory, and attention bias following acute tryptophan depletion and stress exposure. Psychopharmacology (Berl ) 2009;203(4):805-18.
- Greathouse SM, Kovera MB. Instruction bias and lineup presentation moderate the effects of administrator knowledge on eyewitness identification. Law Hum Behav 2009;33(1):70-82.
- Gregory AM, Caspi A, Moffitt TE, Milne BJ, Poulton R, Sears MR. Links between anxiety and allergies: psychobiological reality or possible methodological bias? J Pers 2009;77(2):347-62.
- Gurusamy KS, Gluud C, Nikolova D, Davidson BR. Assessment of risk of bias in randomized clinical trials in surgery. Br J Surg 2009;96(4):342-9.
- Harmer CJ, O'Sullivan U, Favaron E, Massey-Chase R, Ayres R, Reinecke A, et al. Effect of acute antidepressant administration on negative affective bias in depressed patients. Am J Psychiatry 2009;166(10):1178-84.
- Hartling L, Ospina M, Liang Y, Dryden DM, Hooton N, Krebs SJ, et al. Risk of bias versus quality assessment of randomised controlled trials: cross sectional study. BMJ 2009;339:b4012, 2009.
- Kerestes R, Labuschagne I, Croft RJ, O'Neill BV, Bhagwagar Z, Phan KL, et al. Evidence for modulation of facial emotional processing bias during emotional expression decoding by serotonergic and noradrenergic antidepressants: an event-related potential (ERP) study. Psychopharmacology (Berl ) 2009;202(4):621-34.
- Koretz RL. Probiotics, critical illness, and methodologic bias. Nutr Clin Pract 2009;24(1):45-9.
- Lamm E. Conceptual and methodological biases in network models. Ann N Y Acad Sci 2009;1178:291-304, 2009 Oct.:-304.
- Miskowiak KW, Favaron E, Hafizi S, Inkster B, Goodwin GM, Cowen PJ, et al. Effects of erythropoietin on emotional processing biases in patients with major depression: an exploratory fMRI study. Psychopharmacology (Berl ) 2009;207(1):133-42.
- Mohammed MA, Deeks JJ, Girling A, Rudge G, Carmalt M, Stevens AJ, et al. Evidence of methodological bias in hospital standardised mortality ratios: retrospective database study of English hospitals. BMJ 2009;338:b780, 2009.
- Parker RA. Studies should be controlled, randomized, and blinded. Clin Pharmacol Ther 2009;85(5):461-3
- Rosenheck RA, Davis VG, Davis SM, Stroup S, McEvoy J, Swartz M, et al. Can a nonequivalent choice of dosing regimen bias the results of flexible dose double blind trials? The CATIE schizophrenia trial. Schizophr Res 2009;113(1):12-8.
- Sherlaw-Johnson C, Harvey D, Bishop N, Wood H, Hamblin R, Ellis N, et al. Standardised mortality ratios. Methodological bias. BMJ 2009;338:b1746, 2009
- Spazierer D, Skvara H, Dawid M, Fallahi N, Gruber K, Rose K, et al. T helper 2 biased de novo immune response to Keyhole Limpet Hemocyanin in humans. Clin Exp Allergy 2009;39(7):999-1008.
- Ventegodt S, Andersen NJ, Brom B, Merrick J, Greydanus DE. Evidence-based medicine: four fundamental problems with the randomized clinical trial (RCT) used to document chemical medicine. Int J Adolesc Med Health 2009;21(4):485-96.
- Angell M. Industry-sponsored clinical research: a broken system. JAMA 2008;300(9):1069-71.
- Crossley NA, Sena E, Goehler J, Horn J, van der Worp B, Bath PM, et al. Empirical evidence of bias in the design of experimental stroke studies: a metaepidemiologic approach. Stroke 2008;39(3):929-34.
- Decaudin D, Dolcetti R, de CP, Ponzoni M, Vincent-Salomon A, Doglioni C, et al. Variable association between Chlamydophila psittaci infection and ocular adnexal lymphomas: methodological biases or true geographical variations? Anticancer Drugs 2008;19(8):
- Fenwick J, Needleman IG, Moles DR. The effect of bias on the magnitude of clinical outcomes in periodontology: a pilot study. J Clin Periodontol 2008;35(9):775-82.
- Furberg CD, Soliman EZ. Double-blindness protects scientific validity. J Thromb Haemost 2008;6(2):230-1.
- Gerhard T. Bias: considerations for research practice. Am J Health-Syst Pharm 2008;65(22):2159-68.
- Lundh A, Gotzsche PC. Recommendations by Cochrane Review Groups for assessment of the risk of bias in studies. BMC Med Res Methodol 2008;8:22, 2008.
- Paradis C. Bias in surgical research. Ann Surg 2008;248(2):180-8.
- Peinemann F, McGauran N, Sauerland S, Lange S. Negative pressure wound therapy: potential publication bias caused by lack of access to unpublished study results data. BMC Med Res Methodol 2008;8:4
- Rzetelny A, Gilbert DG, Hammersley J, Radtke R, Rabinovich NE, Small SL. Nicotine decreases attentional bias to negative-affect-related Stroop words among smokers. Nicotine Tob Res 2008;10(6):1029-36.
- Schnoll RA, Epstein L, Audrain J, Niaura R, Hawk L, Shields PG, et al. Can the blind see? Participant guess about treatment arm assignment may influence outcome in a clinical trial of bupropion for smoking cessation. J Subst Abuse Treat 2008;34(2):234-41.
- van WG, Mattern C, Verkes RJ, Buitelaar J, Fernandez G. Testosterone biases automatic memory processes in women towards potential mates. Neuroimage 2008;43(1):114-20.
- WaxmanAS, Robinson DA, Evans RB, Hulse DA, Innes JF, Conzemius MG. Relationship between objective and subjective assessment of limb function in normal dogs with an experimentally induced lameness. Vet Surg 2008;37(3):241-6.
- Wood L, Egger M, Gluud LL, Schulz KF, Juni P, Altman DG, et al. Empirical evidence of bias in treatment effect estimates in controlled trials with different interventions and outcomes: meta-epidemiological study. BMJ 2008;336(7644):601-5.
- Zahl PH, Jorgensen KJ, Maehlen J, Gotzsche PC. Biases in estimates of overdetection due to mammography screening. Lancet Oncol 2008;9(3):199-201
- Zhang X, Shu XO, Chow WH, Yang G, Li H, Gao J, et al. Body mass index at various ages and mortality in Chinese women: impact of potential methodological biases. Int J Obes (Lond) 2008;32(7):1130-6
- Gluud C, Gluud LL. Evidence based diagnostics. BMJ 2005;330(7493):724-6.
- Hahn S, Puffer S, Torgerson DJ, Watson J. Methodological bias in cluster randomised trials. BMC Med Res Methodol 2005; 5(1): 10.
- Pildal J, Chan AW, Hrobjartsson A, Forfang E, Altman DG, Gotzsche PC. Comparison of descriptions of allocation concealment in trial protocols and the published reports: cohort study. BMJ 2005; 330(7499): 1049.
- Pildal J, Hróbjartsson A, Jørgensen JK, Hilden J, Altman DG, Gøtzsche PC. Impact of allocation concealment on conclusions drawn from meta-analyses of randomised trials. 2007; International Journal of Epidemiology. [Epub ahead of print] doi:10.1093/ije/dym087
- Schulz KF, Chalmers I, Grimes DA, Altman DG. Assessing the quality of randomization from reports of controlled trials published in obstetrics and gynecology journals. JAMA 1994; 272(2): 125-8.
- Schulz KF, Chalmers I, Altman DG, Grimes DA, Dore CJ. The methodologic quality of randomization as assessed from reports of trials in specialist and general medical journals. Online Journal of Current Clinical Trials 1995; Doc No 197: 81.
- Schulz KF, Chalmers I, Hayes RJ, Altman DG. Empirical evidence of bias. Dimensions of methodological quality associated with estimates of treatment effects in controlled trials. JAMA 1995; 273(5): 408-12.
- Schulz KF, Grimes DA, Altman DG, Hayes RJ. Blinding and exclusions after allocation in randomised controlled trials: survey of published parallel group trials in obstetrics and gynaecology. BMJ 1996; 312(7033): 742-4.
- Siegfried N, Clarke M, Volmink J, Van der Merwe L. African HIV/AIDS trials are more likely to report adequate allocation concealment and random generation than North American trials. PLoS ONE 2008; 3(10):e3491.
- Sterne JA, Juni P, Schulz KF, Altman DG, Bartlett C, Egger M. Statistical methods for assessing the influence of study characteristics on treatment effects in 'meta-epidemiological' research. Statistics in Medicine 2002; 21(11): 1513-24.
- Sutton AJ, Turner EH, Moreno SG. Concerns about reported rank-order of antidepressant efficacy. (Accepted for publication) The Lancet 2009.
- Tierney JF, Stewart LA. Investigating patient exclusion bias in meta-analysis. International Journal of Epidemiology 2005; 34(1): 79-87.
- Wood L. The epidemiology of bias in randomised (clinical) controlled trials: a meta-epidemiological study. PhD thesis. 2006.
- Zhaoxiang Bian, Taixiang Wu, Youping Li, Guanjian Liu, David Moher: Randomized trials published in partial Chinese journals: How many are randomized? Accepted by Trials.
- Senn S. Misunderstanding publication bias: editors are not blameless after all. F1000Res 2012;1:59. [PMID: 24358828]
- Mills EJ, Kanters S, Thorlund. The effects of excluding treatments from network meta-analyses: survey. BMJ 2013;347:f5195. [PMID: 24009242]
- Redmond S, von Elm. Cohort study of trials submitted to ethics committee identified discrepant reporting of outcomes in publications. J Clin Epidemiol 2013 Dec;66(12):1367-75. [PMID: 24075598]
- Wager E, Williams. "Hardly worth the effort"? Medical journals' policies and their editors' and publishers' views on trial registration and publication bias: quantitative and qualitative study. BMJ 2013;347:f5248. [PMID: 24014339]
- Sando IC, Malay S, Chung KC. Analysis of publication bias in the literature for distal radius fracture. [Review]. J Hand Surg [Am] 2013 May;38(5):927-34. [PMID: 23566720]
- Turner EH. Publication bias, with a focus on psychiatry: causes and solutions. [Review]. CNS Drugs 2013 Jun;27(6):457-68. [PMID: 23696308]
- Portalupi S, von Elm. Protocol for a systematic review on the extent of non-publication of research studies and associated study characteristics. Syst 2013;2:2. [PMID: 23302739]
- McDonagh MS. US Food and Drug Administration documents can provide unpublished evidence relevant to systematic reviews. [Review]. J Clin Epidemiol 2013 Oct;66(10):1071-81. [PMID: 23856190]
- de WJ. Why selective publication of statistically significant results can be effective. PLoS ONE 2013;8(6):e66463. [PMID: 23840479]
- Loureiro LV. Is there publication bias towards Brazilian articles on cancer? Einstein 2013 Jan;11(1):15-22. [PMID: 23579739]
- Tonia T. Cancer, meta-analysis and reporting biases: the case of erythropoiesis-stimulating agents. [Review]. Swiss Med Wkly 2013;143:w13776. [PMID: 23740272]
- Leggett NC, Thomas NA, Loetscher. The life of p: "just significant" results are on the rise. Q J Exp Psychol (Colchester ) 2013;66(12):2303-9. [PMID: 24205936]van LM. Recommendations for a uniform assessment of publication bias related to funding source. BMC Med Res Methodol 2013;13:120. [PMID: 24079325]
- Jupiter DC. Accentuate the positive? The negative role of bias. J Foot Ankle Surg 2013 Sep;52(5):690-1. [PMID: 23759211]
- Kicinski M. Publication bias in recent meta-analyses.[Erratum appears in PLoS One. 2014;9(1). doi:10.1371/annotation/a65c0f61-eb99-42f0-828b-5a8662bce4f7]. PLoS ONE 2013;8(11):e81823. [PMID: 24363797]
- Smulders YM. A two-step manuscript submission process can reduce publication bias. J Clin Epidemiol 2013 Sep;66(9):946-7. [PMID: 23845183]
- David SP, Ware JJ, Chu IM, Loftus PD, Fusar-Poli. Potential reporting bias in fMRI studies of the brain. PLoS ONE 2013;8(7):e70104. [PMID: 23936149]
- Lu Y. Mapping publication status and exploring hotspots in a research field: chronic disease self-management. J Adv Nurs 2014 Aug;70(8):1837-44. [PMID: 24438056]
- van LM. Role of editorial and peer review processes in publication bias: analysis of drug trials submitted to eight medical journals. PLoS ONE 2014;9(8):e104846. [PMID: 25118182]
- Simonsohn U. P-curve: a key to the file-drawer. J Exp Psychol Gen 2014 Apr;143(2):534-47. [PMID: 23855496]
- Paucity of negative clinical trials reports and publication bias. J Can Acad Child Adolesc Psychiatry 2014 Feb;Acade.mie canadienne de psychiatrie de l.enfant et de l.adolescent. 23(1):7. [PMID: 24516471]
- Mavridis D. How to assess publication bias: funnel plot, trim-and-fill method and selection models. Evid Based Ment Health 2014 Feb;17(1):30. [PMID: 24477535]
- Burkner PC. Testing for publication bias in diagnostic meta-analysis: a simulation study. Stat Med 2014 Aug 15;33(18):3061-77. [PMID: 24753050]
- Carter EC. Publication bias and the limited strength model of self-control: has the evidence for ego depletion been overestimated? Front Psychol 2014;5:823. [PMID: 25126083]
- van Enst WA, Ochodo E, Scholten RJ, Hooft. Investigation of publication bias in meta-analyses of diagnostic test accuracy: a meta-epidemiological study. BMC Med Res Methodol 2014;14:70. [PMID: 24884381]
- Mavridis D. Exploring and accounting for publication bias in mental health: a brief overview of methods. [Review]. Evid Based Ment Health 2014 Feb;17(1):11-5. [PMID: 24477532]
- Ioannidis JP, Munafo MR, Fusar-Poli. Publication and other reporting biases in cognitive sciences: detection, prevalence, and prevention. [Review]. Trends Cogn Sci 2014 May;18(5):235-41. [PMID: 24656991]
- Jin ZC. A modified regression method to test publication bias in meta-analyses with binary outcomes. BMC Med Res Methodol 2014;14:132. [PMID: 25516509]
- Chan AW. Increasing value and reducing waste: addressing inaccessible research. Lancet 2014 Jan 18;383(9913):257-66. [PMID: 24411650]
- Doucouliagos H. Publication selection and the income elasticity of the value of a statistical life. J Health Econ 2014 Jan;33:67-75. [PMID: 24300998]
- Sham E, Smith. Publication bias in studies of an applied behavior-analytic intervention: an initial analysis. [Review]. J Appl Behav Anal 2014;47(3):663-78. [PMID: 24990802]
- Dal-Re R. Time to ensure that clinical trial appropriate results are actually published. Eur J Clin Pharmacol 2014 Apr;70(4):491-3. [PMID: 24413686]
- Mavridis, D. How to assess publication bias: funnel plot, trim-and-fill method and selection models. Evid Based Ment Health 17 (1):30, 2014.
- Onishi A. Publication bias is underreported in systematic reviews published in high-impact-factor journals: metaepidemiologic study. [Review]. J Clin Epidemiol 2014 Dec;67(12):1320-6. [PMID: 25194857]
- DeLong MR, Hughes DB, Blau JA, Hollenbeck ST. Publication Bias and the Under-Reporting of Complications in the Literature: Have We Dug Our Own Pay-for-Performance Grave? Plast Reconstr Surg 2014 Oct;134(4 Suppl 1):42-3. [PMID: 25254732]
- Franco A. Social science. Publication bias in the social sciences: unlocking the file drawer. Science 2014 Sep;345(6203):1502-5. [PMID: 25170047]
- Gjerdevik M. Improving the error rates of the Begg and Mazumdar test for publication bias in fixed effects meta-analysis. BMC Med Res Methodol 2014;14:109. [PMID: 25245217]
- Papageorgiou SN, Papadopoulos MA, Athanasiou AE. Assessing small study effects and publication bias in orthodontic meta-analyses: a meta-epidemiological study. Clin Oral Investig 2014 May;18(4):1031-44. [PMID: 24526347]
- Miller KA, Bell TP, Germano JM. Understanding publication bias in reintroduction biology by assessing translocations of New Zealand's herpetofauna. Conserv 2014 Aug;28(4):1045-56. [PMID: 24606604]
- Hunter JP. In meta-analyses of proportion studies, funnel plots were found to be an inaccurate method of assessing publication bias. J Clin Epidemiol 2014 Aug;67(8):897-903. [PMID: 24794697]
- Malicki M. Is there a solution to publication bias? Researchers call for changes in dissemination of clinical research results. J Clin Epidemiol 2014 Oct;67(10):1103-10. [PMID: 25034197]
- Khan NA. Randomized controlled trials of rheumatoid arthritis registered at ClinicalTrials.gov: what gets published and when. Arthritis rheumatol 2014 Oct;66(10):2664-74. [PMID: 25154608]
- Moustgaard H. Subjective and objective outcomes in randomized clinical trials: definitions differed in methods publications and were often absent from trial reports. [Review]. J Clin Epidemiol 2014 Dec;67(12):1327-34. [PMID: 25263546]
- Kasenda B. Prevalence, characteristics, and publication of discontinued randomized trials. JAMA 2014 Mar 12;311(10):1045-51. [PMID: 24618966]
- Kuhberger A. Publication bias in psychology: a diagnosis based on the correlation between effect size and sample size. PLoS ONE 2014;9(9):e105825. [PMID: 25192357]
- Mavridis D. A selection model for accounting for publication bias in a full network meta-analysis. Stat Med 2014 Dec 30;33(30):5399-412. [PMID: 25316006]
- Kien C. Barriers to and facilitators of interventions to counter publication bias: thematic analysis of scholarly articles and stakeholder interviews. BMC Health Serv Res 2014;14:551. [PMID: 25719959]
- Papageorgiou SN. Publication bias & small-study effects in pediatric dentistry meta-analyses. The Journal of Evidencebased Dental Practice 2015 Mar;15(1):8-24. [PMID: 25666576]
- Emdin C. Association of cardiovascular trial registration with positive study findings: Epidemiological Study of Randomized Trials (ESORT). JAMA Intern Med 2015 Feb;175(2):304-7. [PMID: 25545611]
- Anglemyer AT. Industry sponsorship and publication bias among animal studies evaluating the effects of statins on atherosclerosis and bone outcomes: a meta-analysis. BMC Med Res Methodol 2015;15(1):12. [PMID: 25880564]
- de BA. Cognitive advantage in bilingualism: an example of publication bias? Psychol Sci 2015 Jan;26(1):99-107. [PMID: 25475825]
- Jin ZC, Zhou XH, He. Statistical methods for dealing with publication bias in meta-analysis. Stat Med 2015 Jan 30;34(2):343-60. [PMID: 25363575]
- Muller KF, Briel M, D'Amario A, Kleijnen J, Marusic A, Wager E, et al. Defining publication bias: protocol for a systematic review of highly cited articles and proposal for a new framework. Syst 2013;2:34. [PMID: 23692820]
- Wager E, Williams P. "Hardly worth the effort"? Medical journals' policies and their editors' and publishers' views on trial registration and publication bias: quantitative and qualitative study. BMJ 2013;347:f5248. [PMID: 24014339]
- Mills EJ, Kanters S, Thorlund K, Chaimani A, Veroniki AA, Ioannidis JP. The effects of excluding treatments from network meta-analyses: survey. BMJ 2013;347:f5195. [PMID: 24009242]
- Mueller KF, Meerpohl JJ, Briel M, Antes G, von EE, Lang B, et al. Detecting, quantifying and adjusting for publication bias in meta-analyses: protocol of a systematic review on methods. Syst Rev 2013;2:60. [PMID: 23885765]
- Guo SW, Evers JL. Lack of transparency of clinical trials on endometriosis. Obstet Gynecol 2013 Jun;121(6):1281-90. [PMID: 23812463]
- Fu R, Selph S, McDonagh M, Peterson K, Tiwari A, Chou R, et al. Effectiveness and harms of recombinant human bone morphogenetic protein-2 in spine fusion: a systematic review and meta-analysis. Ann Intern Med 2013 Jun 18;158(12):890-902. [PMID: 23778906]
- Scherer RW, Huynh L, Ervin AM, Taylor J, Dickersin K. ClinicalTrials.gov registration can supplement information in abstracts for systematic reviews: a comparison study. BMC Med Res Methodol 2013;13:79. [PMID: 23773868]
- Schmucker C, Bluemle A, Briel M, Portalupi S, Lang B, Motschall E, et al. A protocol for a systematic review on the impact of unpublished studies and studies published in the gray literature in meta-analyses. Syst Rev 2013;2:24. [PMID: 23634657]
- Briel M, Muller KF, Meerpohl JJ, von EE, Lang B, Motschall E, et al. Publication bias in animal research: a systematic review protocol. Syst Rev 2013;2:23. [PMID: 23621910]
- Yu IT, Tse SL. Workshop 11-sources of bias in studies of systematic reviews with or without meta-analysis. Hong Kong Med J 2013 Apr;19(2):156-8. [PMID: 23535676]
- Tzoulaki I, Siontis KC, Evangelou E, Ioannidis JP. Bias in associations of emerging biomarkers with cardiovascular disease. JAMA Intern Med 2013 Apr 22;173(8):664-71. [PMID: 23529078]
- Munder T, Brutsch O, Leonhart R, Gerger H, Barth J. Researcher allegiance in psychotherapy outcome research: an overview of reviews. Clin Psychol Rev 2013 Jun;33(4):501-11. [PMID: 23500154]
- Salami K, Alkayed K. Publication bias in pediatric hematology and oncology: analysis of abstracts presented at the annual meeting of the American Society of Pediatric Hematology and Oncology. Pediatr Hematol Oncol 2013 Apr;30(3):165-9. [PMID: 23461713]
- Richards SM, Burrett JA. A proposal for reducing the effect of one of many causes of publication bias. Trials 2013;14:41. [PMID: 23402474]
- Sune P, Sune JM, Montoro JB. Positive outcomes influence the rate and time to publication, but not the impact factor of publications of clinical trial results. PLoS ONE 2013;8(1):e54583. [PMID: 23382919]
- Jannot AS, Agoritsas T, Gayet-Ageron A, Perneger TV. Citation bias favoring statistically significant studies was present in medical research. J Clin Epidemiol 2013 Mar;66(3):296-301. [PMID: 23347853]
- Niemeyer H, Musch J, Pietrowsky R. Publication bias in meta-analyses of the efficacy of psychotherapeutic interventions for depression. J Consult Clin Psychol 2013 Feb;81(1):58-74. [PMID: 23244368]
- Vawdrey DK, Hripcsak G. Publication bias in clinical trials of electronic health records. J Biomed Inform 2013 Feb;46(1):139-41. [PMID: 22975314]
- Richards SM, Burrett JA. A proposal for reducing the effect of one of many causes of publication bias. Trials 2013;14(1):41
- Ahmed I, Sutton AJ, Riley RD. Assessment of publication bias, selection bias, and unavailable data in meta-analyses using individual participant data: a database survey. BMJ 2012;344:d7762.
- Bartels RH, Delye H, Boogaarts J. Financial disclosures of authors involved in spine research: an underestimated source of bias. Eur Spine J 2012;21(7):1229-33.
- Carrasco M, Volkmar FR, Bloch MH. Pharmacologic treatment of repetitive behaviors in autism spectrum disorders: evidence of publication bias. Pediatrics 2012;129(5):e1301-e1310
- De Oliveira GSJ, Chang R,KendallMC, Fitzgerald PC, McCarthy RJ. Publication bias in the anesthesiology literature. Anesth Analg 2012;114(5):1042-8.
- Goodyear-Smith FA, van Driel ML, Arroll B, Del MC. Analysis of decisions made in meta-analyses of depression screening and the risk of confirmation bias: a case study. BMC Med Res Methodol 2012;12:76.
- Hart B, Lundh A, Bero L. Effect of reporting bias on meta-analyses of drug trials: reanalysis of meta-analyses. BMJ 2012;344:d7202.
- Okike K, Kocher MS, Nwachukwu BU, Mehlman CT, Heckman JD, Bhandari M. The fate of manuscripts rejected by The Journal of Bone and Joint Surgery (American Volume). J Bone Joint Surg Am 2012;94(17):e130.
- Rodwin MA, Abramson JD. Clinical trial data as a public good. JAMA 2012;308(9):871-2.
- Rossignol DA, Frye RE. A review of research trends in physiological abnormalities in autism spectrum disorders: immune dysregulation, inflammation, oxidative stress, mitochondrial dysfunction and environmental toxicant exposures. Mol Psychiatry 2012;17(4)
- Scherer RW, Sieving PC, Ervin AM, Dickersin K. Can we depend on investigators to identify and register randomized controlled trials? PLoS One 2012;7(9):e44183.
- Thombs BD, Razykov I. A solution to inappropriate self-citation via peer review. CMAJ 2012;184(16):1864.
- Trinquart L, Abbe A, Ravaud P. Impact of reporting bias in network meta-analysis of antidepressant placebo-controlled trials. PLoS One 2012;7(4):e35219
- Turner EH, Knoepflmacher D, Shapley L. Publication bias in antipsychotic trials: an analysis of efficacy comparing the published literature to the US Food and Drug Administration database. PLoS Med 2012;9(3):e1001189
- Carneiro AV. Bias in clinical studies. Rev Port Cardiol 2011;30(2):235-42.
- Cornell A, Bushman B, Womack K. Analysis of journals that did not meet selection criteria for inclusion in the National Library of Medicine collection but have manuscripts in PubMed Central. J Med Libr Assoc 2011;99(2):168-70.
- Dickersin K, Chalmers I. Recognizing, investigating and dealing with incomplete and biased reporting of clinical research: from Francis Bacon to the WHO. J R Soc Med 2011;104(12):532-8.
- Hrobjartsson A, Kaptchuk TJ, Miller FG. Placebo effect studies are susceptible to response bias and to other types of biases. J Clin Epidemiol 2011;64(11):1223-9.
- Ioannidis JP. Excess significance bias in the literature on brain volume abnormalities. Arch Gen Psychiatry 2011;68(8):773-80
- Kanaan Z, Galandiuk S, Abby M,ShannonKV, Dajani D, Hicks N, et al. The value of lesser-impact-factor surgical journals as a source of negative and inconclusive outcomes reporting. Ann Surg 2011;253(3):619-23.
- Koog YH, We SR, Min BI. Three-armed trials including placebo and no-treatment groups may be subject to publication bias: systematic review. PLoS One 2011;6(5):e20679.
- Koren G, Nickel C. Perpetuating fears: bias against the null hypothesis in fetal safety of drugs as expressed in scientific citations. J Popul Ther Clin Pharmacol 2011;18(1):e28-e32.
- Liu Y, Yang S, Dai J, Xu Y, Zhang R, Jiang H, et al. Risk of bias tool in systematic reviews/meta-analyses of acupuncture in Chinese journals. PLoS One 2011;6(12):e28130
- Moreno SG, Sutton AJ, Ades AE, Cooper NJ, Abrams KR. Adjusting for publication biases across similar interventions performed well when compared with gold standard data. J Clin Epidemiol 2011;64(11):1230-41
- Parekh-Bhurke S, Kwok CS, Pang C, Hooper L,LokeYK, Ryder JJ, et al. Uptake of methods to deal with publication bias in systematic reviews has increased over time, but there is still much scope for improvement. J Clin Epidemiol 2011;64(4):349-57.
- Pfeiffer T, Bertram L, Ioannidis JP. Quantifying selective reporting and the Proteus phenomenon for multiple datasets with similar bias. PLoS One 2011;6(3):e18362.
- Polyzos NP, Valachis A, Mauri D, Ioannidis JP. Industry involvement and baseline assumptions of cost-effectiveness analyses: diagnostic accuracy of the Papanicolaou test. CMAJ 2011;183(6):E337-E343
- Polyzos NP, Valachis A, Patavoukas E, Papanikolaou EG, Messinis IE,TarlatzisBC, et al. Publication bias in reproductive medicine: from the European Society of Human Reproduction and Embryology annual meeting to publication. Hum Reprod 2011;26(6):1371-6.
- Reyes MM, Panza KE, Martin A, Bloch MH. Time-lag bias in trials of pediatric antidepressants: a systematic review and meta-analysis. J Am Acad Child Adolesc Psychiatry 2011;50(1):63-72.
- Smyth RM, Kirkham JJ, Jacoby A, Altman DG, Gamble C, Williamson PR. Frequency and reasons for outcome reporting bias in clinical trials: interviews with trialists. BMJ 2011;342:c7153
- Steen RG. Retractions in the scientific literature: is the incidence of research fraud increasing? J Med Ethics 2011;37(4):249-53.
- Valachis A, Mauri D, Neophytou C, Polyzos NP, Tsali L, Garras A, et al. Translational medicine and reliability of single-nucleotide polymorphism studies: can we believe in SNP reports or not? Int J Med Sci 2011;8(6):492-500
- Bjork BC, Welling P, Laakso M, Majlender P, Hedlund T, Gudnason G. Open access to the scientific journal literature: situation 2009. PLoS One 2010;5(6):e11273.
- Bornmann L, Daniel HD. Do author-suggested reviewers rate submissions more favorably than editor-suggested reviewers? A study on atmospheric chemistry and physics. PLoS One 2010;5(10):e13345
- Bourgeois FT, Murthy S, Mandl KD. Outcome reporting among drug trials registered in ClinicalTrials.gov. Ann Intern Med 2010;153(3):158-66.
- Bowden J,JacksonD, Thompson SG. Modelling multiple sources of dissemination bias in meta-analysis. Stat Med 2010;29(7-8):945-55.
- Cope MB, Allison DB. White hat bias: examples of its presence in obesity research and a call for renewed commitment to faithfulness in research reporting. Int J Obes (Lond) 2010;34(1):84-8
- Crawford JM, Briggs CL, Engeland CG. Publication bias and its implications for evidence-based clinical decision making. J Dent Educ 2010;74(6):593-600.
- Cuijpers P, Smit F, Bohlmeijer E, Hollon SD, Andersson G. Efficacy of cognitive-behavioural therapy and other psychological treatments for adult depression: meta-analytic study of publication bias. Br J Psychiatry 2010;196(3):173-8.
- Dwan K, Gamble C, Kolamunnage-Dona R, Mohammed S, Powell C, Williamson PR. Assessing the potential for outcome reporting bias in a review: a tutorial. Trials 2010;11:52
- Emerson GB, Warme WJ, Wolf FM, Heckman JD, Brand RA, Leopold SS. Testing for the presence of positive-outcome bias in peer review: a randomized controlled trial. Arch Intern Med 2010;170(21):1934-9.
- Fanelli D. Do pressures to publish increase scientists' bias? An empirical support from US States Data. PLoS One 2010;5(4):e10271.
- Garattini L, Koleva D, Casadei G. Modeling in pharmacoeconomic studies: funding sources and outcomes. Int J Technol Assess Health Care 2010;26(3):330-3.
- Gibson LM, Brazzelli M, Thomas BM, Sandercock PA. A systematic review of clinical trials of pharmacological interventions for acute ischaemic stroke (1955-2008) that were completed, but not published in full. Trials 2010;11:43.
- Golder S,LokeYK, Bland M. Unpublished data can be of value in systematic reviews of adverse effects: methodological overview. J Clin Epidemiol 2010;63(10):1071-81.
- Harris P, Takeda A, Loveman E, Hartwell D. Time to full publication of studies of anticancer drugs for breast cancer, and the potential for publication bias. Int J Technol Assess Health Care 2010;26(1):110-6.
- Hemingway H, Philipson P, Chen R, Fitzpatrick NK, Damant J, Shipley M, et al. Evaluating the quality of research into a single prognostic biomarker: a systematic review and meta-analysis of 83 studies of C-Reactive protein in stable coronary artery disease. PLoS Medicine 2010;7(6):e1000286.
- Henmi M, Copas JB. Confidence intervals for random effects meta-analysis and robustness to publication bias. Stat Med 2010;29(29):2969-83.
- Jong JP, Ter RG, Willems DL. Two prognostic indicators of the publication rate of clinical studies were available during ethical review. J Clin Epidemiol 2010;63(12):1342-50.
- Lundh A, Barbateskovic M, Hrobjartsson A, Gotzsche PC. Conflicts of interest at medical journals: the influence of industry-supported randomised trials on journal impact factors and revenue - cohort study. PLoS Med 2010;7(10):e1000354, 2010.
- McGauran N, Wieseler B, Kreis J, Schuler YB, Kolsch H, Kaiser T. Reporting bias in medical research - a narrative review. Trials 2010;11:37.
- Palla L, Higgins JP,WarehamNJ, Sharp SJ. Challenges in the use of literature-based meta-analysis to examine gene-environment interactions. Am J Epidemiol 2010;171(11):1225-32.
- Pitak-Arnnop P, Sader R, Rapidis AD, Dhanuthai K, Bauer U, Herve C, et al. Publication bias in oral and maxillofacial surgery journals: an observation on published controlled trials. Journal of Cranio maxillo facial Surgery 2010;38(1):4-10.
- Ponce FA, Lozano AM. Highly cited works in neurosurgery. Part I: the 100 top-cited papers in neurosurgical journals. J Neurosurg 2010;112(2):223-32.
- Schlaepfer TE, Fins JJ. Deep brain stimulation and the neuroethics of responsible publishing: when one is not enough. JAMA 2010;303(8):775-6.
- Schwarzer G, Carpenter J, Rucker G. Empirical evaluation suggests Copas selection model preferable to trim-and-fill method for selection bias in meta-analysis. J Clin Epidemiol 2010;63(3):282-8.
- Song F, Parekh S, Hooper L,LokeYK, Ryder J, Sutton AJ, et al. Dissemination and publication of research findings: an updated review of related biases. Health Technol Assess 2010;14(8):iii, ix-iii,193
- Soonawala D, Middelburg RA, Egger M, Vandenbroucke JP, Dekkers OM. Efficacy of experimental treatments compared with standard treatments in non-inferiority trials: a meta-analysis of randomized controlled trials. Int J Epidemiol 2010;39(6):1567-81.
- Tan PCF, Graham CA. Full text publication rates of studies presented at an Asian emergency medicine scientific meeting.Hong KongJournal of Emergency Medicine 2010;17(2):154-7.
- Whiting PF, Smidt N, Sterne JA, Harbord R,BurtonA, Burke M, et al. Systematic review: accuracy of anti-citrullinated Peptide antibodies for diagnosing rheumatoid arthritis. Ann Intern Med 2010;152(7):456-64.
- Aarssen LW, Lortie CJ, Budden AE, Koricheva J, Leimu R, Tregenza T. Does publication in top-tier journals affect reviewer behavior? PLoS One 2009;4(7):e6283.
- Barker BR.Are positive alternative medical therapy trials credible?: Evidence from four high-impact medical journals. Evaluation and the Health Professions 2009;32(4):349-69.
- Bolac C, Orosco A, Guillet G, Quist D, Derancourt C. [Publication rate for oral presentations made at the Journees Dermatologiques de Paris meeting]. Annales de Dermatologie et de Venereologie 2009;136(1):21-7.
- Booth CM, Maitre A, Ding K, Farn K, Fralick M, Phillips C, et al. Presentation of nonfinal results of randomized controlled trials at major oncology meetings. J Clin Oncol 2009;27(24):3938-44.
- Borm GF, Heijer M, Zielhuis GA. Publication bias was not a good reason to discourage trials with low power. J Clin Epidemiol 2009;62(1):47-53.
- Borm GF, Donders AR. Updating meta-analyses leads to larger type I errors than publication bias. J Clin Epidemiol 2009;62(8):825-30.
- Carpenter JR, Schwarzer G, Rucker G, Kunstler R. Empirical evaluation showed that the Copas selection model provided a useful summary in 80% of meta-analyses. J Clin Epidemiol 2009;62(6):624-31.
- Chalmers I, Glasziou P. Avoidable waste in the production and reporting of research evidence. Obstet Gynecol 2009;114(6):1341-5.
- Decullier E, Chan AW, Chapuis F. Inadequate dissemination of phase I trials: a retrospective cohort study. PLoS Med 2009;6(2):e1000034
- Driel ML, Sutter A, Maeseneer J, Christiaens T. Searching for unpublished trials in Cochrane reviews may not be worth the effort. J Clin Epidemiol 2009;62(8):838-44
- Etter JF, Stapleton J. Citations to trials of nicotine replacement therapy were biased toward positive results and high-impact-factor journals. J Clin Epidemiol 2009;62(8):831-7.
- Gartlehner G, Thieda P, Hansen RA, Morgan LC, Shumate JA, Nissman DB. Inadequate reporting of trials compromises the applicability of systematic reviews. Int J Technol Assess Health Care 2009;25(3):323-30.
- Hammer GP, du Prel JB, Blettner M. Avoiding bias in observational studies: part 8 in a series of articles on evaluation of scientific publications. Dtsch Arztebl Int 2009;106(41):664-8.
- Hoeg RT, Lee JA, Mathiason MA, Rokkones K, Serck SL, Crampton KL, et al. Publication outcomes of phase II oncology clinical trials. Am J Clin Oncol 2009;32(3):253-7.
- Hopewell S, Loudon K, Clarke MJ, Oxman AD, Dickersin K. Publication bias in clinical trials due to statistical significance or direction of trial results. Cochrane Database Syst Rev 2009;(1):MR000006.
- Irwin RS. The role of conflict of interest in reporting of scientific information. Chest 2009;136(1):253-9.
- Korn EL, Freidlin B, Mooney M. Stopping or reporting early for positive results in randomized clinical trials: The National Cancer Institute Cooperative Group experience from 1990 to 2005. J Clin Oncol 2009;27(10):1712-21.
- Lopez-Gonzalez E,HerdeiroMT, Figueiras A. Determinants of under-reporting of adverse drug reactions: a systematic review. Drug Saf 2009;32(1):19-31.
- Mathew SJ, Charney DS. Publication bias and the efficacy of antidepressants. Am J Psychiatry 2009;166(2):140-5.
- Mathieu S, Boutron I, Moher D, Altman DG, Ravaud P. Comparison of registered and published primary outcomes in randomized controlled trials. JAMA 2009;302(9):977-84.
- Matsubayashi M, Kurata K, Sakai Y, Morioka T, Kato S, Mine S, et al. Status of open access in the biomedical field in 2005. J Med Libr Assoc 2009;97(1):4-11.
- McGoey L. Sequestered evidence and the distortion of clinical practice guidelines. Perspectives in Biology & Medicine 2009;52(2):203-17.
- Moreno SG, Sutton AJ, Ades AE, Stanley TD, Abrams KR, Peters JL, et al. Assessment of regression-based methods to adjust for publication bias through a comprehensive simulation study. BMC Med Res Methodol 2009;9:2.
- Moreno SG, Sutton AJ, Turner EH, Abrams KR, Cooper NJ, Palmer TM, et al. Novel methods to deal with publication biases: secondary analysis of antidepressant trials in the FDA trial registry database and related journal publications. BMJ 2009;339:b2981.
- Nieto A, Mazon A, Pamies R, Bruno L, Navarro M, Montanes A. Sublingual immunotherapy for allergic respiratory diseases: an evaluation of meta-analyses. J Allergy Clin Immunol 2009;124(1):157-61.
- Rasmussen N, Lee K, Bero L. Association of trial registration with the results and conclusions of published trials of new oncology drugs. Trials 2009;10:116.
- Rising K, Bacchetti P, Bero L. Correction: Reporting bias in drug trials submitted to the Food and Drug Administration: review of publication and presentation. PLoS Medicine 2009;6(1):e117.
- Ross JS, Mulvey GK, Hines EM, Nissen SE, Krumholz HM. Trial publication after registration in ClinicalTrials.Gov: a cross-sectional analysis. PLoS Medicine 2009;6(9):e1000144.
- Senn SJ. Overstating the evidence: double counting in meta-analysis and related problems. BMC Medical Research Methodology 2009;9:10.
- Slobogean GP, Verma A, Giustini D, Slobogean BL, Mulpuri K. MEDLINE, EMBASE, and Cochrane index most primary studies but not abstracts included in orthopedic meta-analyses. J Clin Epidemiol 2009;62(12):1261-7.
- Song F, Parekh-Bhurke S, Hooper L,LokeYK, Ryder JJ, Sutton AJ, et al. Extent of publication bias in different categories of research cohorts: a meta-analysis of empirical studies. BMC Med Res Methodol 2009;9:79.
- Sridharan L, Greenland P. Editorial policies and publication bias: the importance of negative studies. Arch Intern Med 2009;169(11):1022-3.
- Tfelt-Hansen PC. Unpublished clinical trials with sumatriptan. Lancet 2009;374(9700):1501-2.
- Tricco AC, Pham B, Brehaut J, Tetroe J, Cappelli M, Hopewell S, et al. An international survey indicated that unpublished systematic reviews exist. J Clin Epidemiol 2009;62(6):617-23.
- Tricco AC, Tetzlaff J, Pham B, Brehaut J, Moher D. Non-Cochrane vs. Cochrane reviews were twice as likely to have positive conclusion statements: cross-sectional study. J Clin Epidemiol 2009;62(4):380-6.
- Vecchi S, Belleudi V, Amato L, Davoli M,PerucciCA. Does direction of results of abstracts submitted to scientific conferences on drug addiction predict full publication? BMC Med Res Methodol 2009;9:23.
- Vedula SS, Bero L, Scherer RW, Dickersin K. Outcome reporting in industry-sponsored trials of gabapentin for off-label use. N Engl J Med 2009;361(20):1963-71
- Viereck C, Boudes P. An analysis of current pharmaceutical industry practices for making clinical trial results publicly accessible. Contemporary Clinical Trials 2009;30(4):293-9.
- Wager E, Fiack S, Graf C, Robinson A, Rowlands I. Science journal editors' views on publication ethics: results of an international survey. J Med Ethics 2009;35(6):348-53.
- Wagner C, Gebremichael MD,TaylorMK, Soltys MJ. Disappearing act: decay of uniform resource locators in health care management journals. J Med Libr Assoc 2009;97(2):122-30.
- Akbari-Kamrani M, Shakiba B, Parsian S. Transition from congress abstract to full publication for clinical trials presented at laser meetings. Lasers in Medical Science 2008;23(3):295-9.
- Angell M. Industry-sponsored clinical research: a broken system. JAMA 2008;300(9):1069-71.
- Bekkering GE, Harris RJ, Thomas S, Mayer AM, Beynon R, Ness AR, et al. How much of the data published in observational studies of the association between diet and prostate or bladder cancer is usable for meta-analysis? Am J Epidemiol 2008;167(9):1017-26.
- Boffetta P, McLaughlin JK, La VC, Tarone RE, Lipworth L, Blot WJ. False-positive results in cancer epidemiology: a plea for epistemological modesty. J Natl Cancer Inst 2008;100(14):988-95
- Boschen MJ. Publication trends in individual anxiety disorders: 1980-2015. J Anxiety Disord 2008;22(3):570-5.
- Bubela T, Boon H, Caulfield T. Herbal remedy clinical trials in the media: a comparison with the coverage of conventional pharmaceuticals. BMC Medicine 2008;6(1):35.
- Chan AW, Hrobjartsson A, Jorgensen KJ, Gotzsche PC, Altman DG. Discrepancies in sample size calculations and data analyses reported in randomised trials: comparison of publications with protocols. BMJ 2008;337:a2299.
- Chan AW. Bias, spin, and misreporting: time for full access to trial protocols and results. PLoS Med 2008;5(11):e230.
- Copas JB, Malley PF. A robust P-value for treatment effect in meta-analysis with publication bias. Stat Med 2008;27(21):4267-78.
- Dwan K, Gamble C, Williamson PR, Altman DG. Reporting of clinical trials: a review of research funders' guidelines. Trials 2008;9:66.
- Dwan K, Altman DG, Arnaiz JA, Bloom J, Chan AW, Cronin E, et al. Systematic review of the empirical evidence of study publication bias and outcome reporting bias. PLoS One 2008;3(8):e3081.
- Elm E, Rollin A, Blumle A, Huwiler K, Witschi M, Egger M. Publication and non-publication of clinical trials: longitudinal study of applications submitted to a research ethics committee. Swiss Med Wkly 2008;138(13-14):197-203.
- Faruqui RA, Ahmer S, Bhatti A, Faruqui A. Psychotherapy and pharmacotherapy randomized controlled trials are equally likely to report a positive outcome. J Pak Med Assoc 2008;58(1):37-8.
- FormannAK. Estimating the proportion of studies missing for meta-analysis due to publication bias. Contemporary Clinical Trials 2008;29(5):732-9.
- Golder S,LokeYK. Is there evidence for biased reporting of published adverse effects data in pharmaceutical industry-funded studies? Br J Clin Pharmacol 2008;66(6):767-73.
- Greenberg D, Wacht O, Pliskin JS. Peer review in publication: factors associated with the full-length publication of studies presented in abstract form at the annual meeting of the Society for Medical Decision Making. Med Decis Making 2008;28(6):938-42.
- Hagen NT. Harmonic allocation of authorship credit: source-level correction of bibliometric bias assures accurate publication and citation analysis. PLoS One 2008;3(12):e4021.
- Harlan WR, Zarin DA, Williams RJ: TT. A public database of clinical trials results: progress and issues [abstract]. Clinical Trials 2008;5(4):392.
- Huss A, Egger M, Hug K, Huwiler-Muntener K, Roosli M. Source of funding and results of studies of health effects of mobile phone use: systematic review of experimental studies. Ciencia & Saude Coletiva 2008;13(3):1005-12.
- Iansavichene AE, Sampson M, McGowan J, Ajiferuke ISY. Should systematic reviewers search for randomised controlled trials published as letters? Ann Intern Med 2008;148(9):714-5.
- Ioannidis JP. Why most discovered true associations are inflated. Epidemiology 2008;19(5):640-8.
- Jureidini JN, McHenry LB, Mansfield PR. Clinical trials and drug promotion: selective reporting of study 329. International Journal of Risk and Safety in Medicine 2008;20(1-2):73-81.
- Kheifets L, Olsen J. Should epidemiologists always publish their results? Yes, almost always. Epidemiology 2008;19(4):532-3.
- Kuriya B, Schneid EC, Bell CM. Quality of pharmaceutical industry press releases based on original research. PLoS One 2008;3(7):e2828
- Lee K, Bacchetti P, Sim I. Publication of clinical trials supporting successful new drug applications: a literature analysis. PLoS Medicine 2008;5(9):e191.
- Mahid SS, Qadan M,HornungCA, Galandiuk S. Assessment of publication bias for the surgeon scientist. Br J Surg 2008;95(8):943-9.
- Matias-Guiu J, Garcia-Ramos R. [The impact factor and editorial decisions]. Neurologia 2008;23(6):342-8
- Metcalfe S, Burgess C, Laking G, Evans J, Wells S, Crausaz S. Trastuzumab: possible publication bias. Lancet 2008;371(9625):1646-8.
- Nassir GS, Shirzadi AA, Filkowski M. Publication bias and the pharmaceutical industry: the case of lamotrigine in bipolar disorder. Medscape J Med 2008;10(9):211.
- Okike K, Kocher MS,Mehlman CT, Heckman JD, Bhandari M. Publication bias in orthopaedic research: an analysis of scientific factors associated with publication in the Journal of Bone and Joint Surgery (American Volume). J Bone Joint Surg Am 2008;90(3):595
- Peinemann F, McGauran N, Sauerland S, Lange S. Negative pressure wound therapy: potential publication bias caused by lack of access to unpublished study results data. BMC Med Res Methodol 2008;8:4
- Peters JL, Sutton AJ, Jones DR, Abrams KR, Rushton L. Contour-enhanced meta-analysis funnel plots help distinguish publication bias from other causes of asymmetry. J Clin Epidemiol 2008;61(10):991-6.
- Petticrew M, Egan M, Thomson H, Hamilton V, Kunkler R, Roberts H. Publication bias in qualitative research: what becomes of qualitative research presented at conferences? Journal of Epidemiology & Community Health 2008;62(6):552-4.
- Ramsey S, Scoggins J. Commentary: practicing on the tip of an information iceberg? Evidence of underpublication of registered clinical trials in oncology. Oncologist 2008;13(9):925-9.
- Resnik DB, Gutierrez-Ford C, Peddada S. Perceptions of ethical problems with scientific journal peer review: an exploratory study. Sci Eng Ethics 2008;14(3):305-10.
- Rising K, Bacchetti P, Bero L. Reporting bias in drug trials submitted to the Food and Drug Administration: review of publication and presentation. PLoS Med 2008;5(11):e217.
- Rucker G, Schwarzer G, Carpenter J. Arcsine test for publication bias in meta-analyses with binary outcomes. Stat Med 2008;27(5):746-63.
- Saveleva E, Selinski S. Meta-analyses with binary outcomes: How many studies need to be omitted to detect a publication bias? Journal of Toxicology and Environmental Health 2008;71(13-14):845-50.
- Shakiba B, Salmasian H, Yousefi-Nooraie R, Rohanizadegan M. Factors influencing editors' decision on acceptance or rejection of manuscripts: the authors' perspective. Archives of Iranian Medicine 2008;11(3):257-62.
- Stringer MJ, Sales-Pardo M,Nunes Amaral LA.Effectiveness of journal ranking schemes as a tool for locating information. PLoS One 2008;3(2):e1683.
- Takeda A, Loveman E, Harris P, Hartwell D, Welch K. Time to full publication of studies of anti-cancer medicines for breast cancer and the potential for publication bias: a short systematic review. Health Technol Assess 2008;12(32):1-46.
- The PLoS Medicine Editors. Next stop, don't block the doors: opening up access to clinical trials results. PLoS Medicine 2008;5(7):e160.
- Trikalinos NA, Evangelou E, Ioannidis JP. Falsified papers in high-impact journals were slow to retract and indistinguishable from nonfraudulent papers. J Clin Epidemiol 2008;61(5):464-70.
- Turner EH, Matthews AM, Linardatos E, Tell RA, Rosenthal R. Selective publication of antidepressant trials and its influence on apparent efficacy. N Engl J Med 2008;358(3):252-60.
- Uysal S, Tuglu B, Ozalp Y, Onvural B. Fate of abstracts presented at the 2002 IFCC meeting. Clin Chem Lab Med 2008;46(11):1562-7.
- Young NS, Ioannidis JP, Al-Ubaydli O. Why current publication practices may distort science. PLoS Medicine 2008;5(10):e201.
- Baker R, Jackson D. Using journal impact factors to correct for the publication bias of medical studies. Biometrics 2006; 62(3): 785-92.
- Copas J, Jackson D. A bound for publication bias based on the fraction of unpublished studies. Biometrics 2004; 60(1): 146-53.
- Copas JB, Shi JQ. A sensitivity analysis for publication bias in systematic reviews. Stat Methods Med Res 2001; 10(4): 251-65.
- Decullier E, Lheritier V, Chapuis F. Fate of biomedical research protocols and publication bias in France: retrospective cohort study. BMJ 2005; 331(7507): 19.
- Decullier E, Chapuis F. Oral presentation bias: a retrospective cohort study. J Epidemiol Community Health 2007; 61(3): 190-3.
- Deeks JJ, Macaskill P, Irwig L. The performance of tests of publication bias and other sample size effects in systematic reviews of diagnostic test accuracy was assessed. J Clin Epidemiol 2005; 58(9): 882-93.
- Dickersin K. The existence of publication bias and risk factors for its occurrence. JAMA 1990; 263(10): 1385-9.
- Dickersin K. How important is publication bias? A synthesis of available data. AIDS Educ Prev 1997; 9(1 Suppl): 15-21.
- Dickersin K, Min YI, Meinert CL. Factors influencing publication of research results. Follow up of applications submitted to two institutional review boards. JAMA 1992;263:374-378.
- Dickersin K, Min YI. NIH clinical trials and publication bias. Online Journal of Current Clinical Trials. 1993. Document No. 50.
- Dickersin K (2004a). Publication bias: recognising the problem, understanding its origins and scope, and preventing harm. In: Rothstein H, Sutton A, Borenstein M, eds. Handbook of publication bias. New York: Wiley.
- Dubben HH, Beck-Bornholdt HP. Systematic review of publication bias in studies on publication bias. BMJ 2005; 331(7514): 433-4.
- Duval S, Tweedie R. Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 2000; 56(2): 455-63.
- Dwan K, Altman DG, Arnaiz JA, Bloom J, Chan A, et al. Systematic review of the empirical evidence of study publication bias and outcome reporting bias. PLos ONE 2008; 3(8): e3081.
- Easterbrook PJ, Berlin JA, Gopalan R, Matthews DR. Publication bias in clinical research. Lancet 1991; 337(8746): 867-72.
- Egger M, Juni P, Bartlett C, Holenstein F, Sterne J. How important are comprehensive literature searches and the assessment of trial quality in systematic reviews? Empirical study. Health Technol Assess 2003; 7(1): 1-76.
- Harbord RM, Egger M, Sterne JA. A modified test for small-study effects in meta-analyses of controlled trials with binary endpoints. Stat Med 2006; 25(20): 3443-57.
- Hartling L, Craig WR, Russell K, Stevens K, Klassen TP. Factors influencing the publication of randomized controlled trials in child health research. Arch Pediatr Adolesc Med 2004; 158(10): 983-7.
- Hopewell S, Clarke M, Stewart L, Tierney J. Time to publication for results of clinical trials. Cochrane Database Syst Rev 2007; (2): MR000011.
- Hopewell S, Mallet S. ?Publication bias in clinical trials due to statistical significance or direction of trial results?. The Cochrane Collaboration. Podcasts from The Cochrane Library, Issue 1, 2009.
- Ioannidis JP, Trikalinos TA. The appropriateness of asymmetry tests for publication bias in meta-analyses: a large survey. CMAJ 2007; 176(8): 1091-6.
- Klassen TP, Wiebe N, Russell K, Stevens K, Hartling L, Craig WR, et al. Abstracts of randomized controlled trials presented at the society for pediatric research meeting: an example of publication bias. Arch Pediatr Adolesc Med 2002; 156(5): 474-9.
- Lau J, Ioannidis JP, Terrin N, Schmid CH, Olkin I. The case of the misleading funnel plot. BMJ 2006; 333(7568): 597-600.
- Macaskill P, Walter SD, Irwig L. A comparison of methods to detect publication bias in meta-analysis. Stat Med 2001; 20(4): 641-54.
- McAuley L, Pham B, Tugwell P, Moher D. Does the inclusion of grey literature influence the estimates of intervention effectiveness reported in meta-analyses? Lancet 2000; 356: 1228-1231.
- Moreno SG, Sutton AJ, Ades AE, Stanley TD, Abrams KR, Peters JL, et al. Assessment of regression-based methods to adjust for publication bias through a comprehensive simulation study. BMC Med Res Methodol 2009;9:2.
- Moreno SG, Sutton AJ, Turner EH, Abrams KR, Cooper NJ, Palmer TM, et al. Novel methods to deal with publication biases: ?global? application to antidepressant trials where a gold standard exists. BMJ (Conditionally accepted for publication) 2009.
- Peters JL, Sutton AJ, Jones DR, Abrams KR, Rushton L. Comparison of two methods to detect publication bias in meta-analysis. JAMA 2006; 295(6): 676-80.
- Pham B, Platt R, McAuley L, Klassen TP, Moher D. Is there a "best" way to detect and minimize publication bias? An empirical evaluation. Eval Health Prof 2001; 24(2): 109-25.
- Raina PS, Brehaut JC, Platt RW, Klassen TP, Moher D, St JP, et al. The influence of display and statistical factors on the interpretation of metaanalysis results by physicians. Medical Care 2005; 43(12): 1242-9.
- Rucker G, Schwarzer G, Carpenter J. Arcsine test for publication bias in meta-analyses with binary outcomes. Stat Med. 2007.
- Schwarzer G, Antes G, Schumacher M. Inflation of type I error rate in two statistical tests for the detection of publication bias in meta-analyses with binary outcomes. Stat Med 2002; 21(17): 2465-77.
- Schwarzer G, Antes G, Schumacher M. A test for publication bias in meta-analysis with sparse binary data. Stat Med 2007; 26(4): 721-33.
- Simes RJ. Publication bias: the case for an international registry of clinical trials. J Clin Oncol 1986; 4(10): 1529-41.
- Smith ML. Publication bias and meta-analysis. Evaluation in Education 1980; 4: 22-4.
- Song F, Khan KS, Dinnes J, Sutton AJ. Asymmetric funnel plots and publication bias in meta-analyses of diagnostic accuracy. International Journal of Epidemiology 2002; 31(1): 88-95.
- Sterling TD. Publication decisions and their possible effects on inferences drawn from tests of significance - or vice versa. J Am Stat Assoc 1959; 54: 30-34
- Stern JM, Simes RJ. Publication bias: evidence of delayed publication in a cohort study of clinical research projects. BMJ 1997; 315(7109): 640-5.
- Sterne JA, Gavaghan D, Egger M. Publication and related bias in meta-analysis: power of statistical tests and prevalence in the literature. J Clin Epidemiol 2000; 53(11): 1119-29.
- Sutton AJ, Song F, Gilbody SM, Abrams KR. Modelling publication bias in meta-analysis: a review. Statistical Methods in Medical Research 2000; 9(5): 421-45.
- Sutton AJ, Duval SJ, Tweedie RL, Abrams KR, Jones DR. Empirical assessment of effect of publication bias on meta-analyses. BMJ 2000; 320(7249 ):1574-7.
- Terrin N, Schmid CH, Lau J, Olkin I. Adjusting for publication bias in the presence of heterogeneity. Stat Med 2003 ;22(13): 2113-26.
- Terrin N, Schmid CH, Lau J. In an empirical evaluation of the funnel plot, researchers could not visually identify publication bias. J Clin Epidemiol 2005; 58(9): 894-901.
- Toma M, McAlister FA, Bialy L, Adams D, Vandermeer B, Armstrong PW. Transition from meeting abstract to full-length journal article for randomized controlled trials. JAMA. 2006; 295 (11): 1281-7.
- Vandenbroucke JP (1988). Passive smoking and lung cancer: a publication bias? BMJ 296:391-2
Selective Outcome Reporting Bias
- Trinquart L. Adjustment for reporting bias in network meta-analysis of antidepressant trials. BMC Med Res Methodol 2012;12:150. [PMID: 23016799]
- Smith SM, Wang AT, Pereira. Discrepancies between registered and published primary outcome specifications in analgesic trials: ACTTION systematic review and recommendations. [Review]. Pain 2013 Dec;154(12):2769-74. [PMID: 23962590]
- Knuppel H. How psychiatry journals support the unbiased translation of clinical research. A cross-sectional study of editorial policies. PLoS ONE 2013;8(10):e75995. [PMID: 24146806]
- Dwan K. Systematic review of the empirical evidence of study publication bias and outcome reporting bias - an updated review. [Review]. PLoS ONE 2013;8(7):e66844. [PMID: 23861749]
- Li XQ, Yang GL, Tao KM, Zhang HQ, Zhou QH, Ling CQ. Comparison of registered and published primary outcomes in randomized controlled trials of gastroenterology and hepatology. Scand J Gastroenterol 2013 Dec;48(12):1474-83. [PMID: 24131272]
- Copas J. A model-based correction for outcome reporting bias in meta-analysis. Biostatistics 2014 Apr;15(2):370-83. [PMID: 24215031]
- Page MJ, McKenzie JE, Kirkham. Bias due to selective inclusion and reporting of outcomes and analyses in systematic reviews of randomised trials of healthcare interventions. Cochrane Database Syst Rev 2014;10:MR000035. [PMID: 25271098]
- Huan LN, Tejani AM, Egan. Biomedical journals lack a consistent method to detect outcome reporting bias: a cross-sectional analysis. J Clin Pharm Ther 2014 Oct;39(5):501-6. [PMID: 24828874]
- Saini P. Selective reporting bias of harm outcomes within studies: findings from a cohort of systematic reviews. BMJ 2014;349:g6501. [PMID: 25416499]
- Wiedermann CJ. Reporting bias in trials of volume resuscitation with hydroxyethyl starch. [Review]. Wien Klin Wochenschr 2014 Apr;126(7-8):189-94. [PMID: 24596076]
- Sedgwick P. Meta-analysis: testing for reporting bias. [Review]. BMJ 2015;350:g7857. [PMID: 25555827]
- Nankervis H, Baibergenova A, Williams HC, Thomas KS. Prospective registration and outcome-reporting bias in randomized controlled trials of eczema treatments: a systematic review. [Review]. J Invest Dermatol 2012 Dec;132(12):2727-34. [PMID: 22832489]
- Liu JP, Han M, Li XX, Mu YJ, Lewith G, Wang YY, et al. Prospective registration, bias risk and outcome-reporting bias in randomised clinical trials of traditional Chinese medicine: an empirical methodological study. BMJ Open 2013;3(7). [PMID: 23864210]
- Johnston BC, Patrick DL, Busse JW, Schunemann HJ, Agarwal A, Guyatt GH. Patient-reported outcomes in meta-analyses--Part 1: assessing risk of bias and combining outcomes. [Review]. Health Qual Life Outcomes 2013;11(1):109. [PMID: 23815754]
- Matthew J. Page, Joanne E. McKenzie, Andrew Forbes, Many scenarios exist for selective inclusion and reporting of results in randomized trials and systematic reviews. Journal of Clinical Epidemiology 2013; 66(5):524-537
- Egan G, Lee J, Minhas R, Tejani AM. Does outcome reporting bias "cause" cancer? Risks associated with hidden data on Angiotensin receptor blockers. Can J Hosp Pharm 2012;65(5):387-93.
- Hartling L, Hamm M, Klassen T, Chan AW, Meremikwu M, Moyer V, et al. Standard 2: containing risk of bias. Pediatrics 2012;129 Suppl 3:S124-31, 2012 Jun.:-31
- Kirkham JJ, Riley RD, Williamson PR. A multivariate meta-analysis approach for reducing the impact of outcome reporting bias in systematic reviews. Stat Med 2012;31(20):2179-95.
- Meerpohl JJ, Langer G, Perleth M, Gartlehner G, Kaminski-Hartenthaler A, Schunemann H. GRADE guidelines: 4. Rating the quality of evidence - limitations of clinical trials (risk of bias). Z 2012;Evid. Fortbild. Qual. Gesundh.wes.. 106(6):457-69.
- Nankervis H, Baibergenova A, Williams HC, Thomas KS. Prospective registration and outcome-reporting bias in randomized controlled trials of eczema treatments: a systematic review. J Invest Dermatol 2012;132(12):2727-34.
- Pearson M, Peters J. Outcome reporting bias in evaluations of public health interventions: evidence of impact and the potential role of a study register. J Epidemiol Community Health 2012;66(4):286-9.
- Turner EH, Knoepflmacher D, Shapley L. Publication bias in antipsychotic trials: an analysis of efficacy comparing the published literature to the US Food and Drug Administration database. PLoS Med 2012;9(3):e1001189
- Djulbegovic B, Kumar A, Magazin A, Schroen AT, Soares H, Hozo I, et al. Optimism bias leads to inconclusive results-an empirical study. J Clin Epidemiol 2011;64(6):583-93
- Dwan K, Altman DG, Cresswell L, Blundell M, Gamble CL, Williamson PR. Comparison of protocols and registry entries to published reports for randomised controlled trials. Cochrane Database Syst Rev 2011;(1):MR000031, 2011.
- Guyatt GH, Oxman AD, Vist G, Kunz R, Brozek J, Alonso-Coello P, et al. GRADE guidelines: 4. Rating the quality of evidence--study limitations (risk of bias). J Clin Epidemiol 2011;64(4):407-15.
- Hartling L, Bond K, Vandermeer B, Seida J, Dryden DM, Rowe BH. Applying the risk of bias tool in a systematic review of combination long-acting beta-agonists and inhaled corticosteroids for persistent asthma. PLoS One 2011;6(2):e17242, 2011.
- Haut ER, Pronovost PJ. Surveillance bias in outcomes reporting. JAMA 2011;305(23):2462-3.
- Howland RH. What you see depends on where you're looking and how you look at it: publication bias and outcome reporting bias. J Psychosoc Nurs Ment Health Serv 2011;49(8):13-5.
- Howland RH. Publication bias and outcome reporting bias: agomelatine as a case example. J Psychosoc Nurs Ment Health Serv 2011;49(9):11-4.
- Hrobjartsson A, Kaptchuk TJ, Miller FG. Placebo effect studies are susceptible to response bias and to other types of biases. J Clin Epidemiol 2011;64(11):1223-9.
- Ioannidis JP. Excess significance bias in the literature on brain volume abnormalities. Arch Gen Psychiatry 2011;68(8):773-80
- Jacobs A. The effect of funding source on outcome reporting among drug trials. Ann Intern Med 2011;154(2):137.
- Kanaan Z, Galandiuk S, Abby M,ShannonKV, Dajani D, Hicks N, et al. The value of lesser-impact-factor surgical journals as a source of negative and inconclusive outcomes reporting. Ann Surg 2011;253(3):619-23.
- Liu Y, Yang S, Dai J, Xu Y, Zhang R, Jiang H, et al. Risk of bias tool in systematic reviews/meta-analyses of acupuncture in Chinese journals. PLoS One 2011;6(12):e28130
- Matthews GA, Dumville JC, Hewitt CE, Torgerson DJ. Retrospective cohort study highlighted outcome reporting bias inUKpublicly funded trials. J Clin Epidemiol 2011;64(12):1317-24.
- Rahn DD, Abed H, Sung VW, Matteson KA, Rogers RG, Morrill MY, et al. Systematic review highlights difficulty interpreting diverse clinical outcomes in abnormal uterine bleeding trials. J Clin Epidemiol 2011;64(3):293-300.
- Simunovic N, Walter S, Devereaux PJ, Sprague S, Guyatt GH, Schemitsch E, et al. Outcomes assessment in the SPRINT multicenter tibial fracture trial: adjudication committee size has trivial effect on trial results. J Clin Epidemiol 2011;64(9):1023-33.
- Smyth RM, Kirkham JJ, Jacoby A, Altman DG, Gamble C, Williamson PR. Frequency and reasons for outcome reporting bias in clinical trials: interviews with trialists. BMJ 2011;342:c7153
- Turner EH. The effect of funding source on outcome reporting among drug trials. Ann Intern Med 2011;154(2):137
- Yuan JC, Shyamsunder N,BaraoVA, Lee DJ, Sukotjo C. Publication bias in five dental implant journals: an observation from 2005 to 2009. Int J Oral Maxillofac Implants 2011;26(5):1024-32.
- Zarin DA, Tse T. The effect of funding source on outcome reporting among drug trials. Ann Intern Med 2011;154(2):137-8.
- Bourgeois FT, Murthy S, Mandl KD. Outcome reporting among drug trials registered in ClinicalTrials.gov. Ann Intern Med 2010;153(3):158-66.
- Boutron I, Dutton S, Ravaud P, Altman DG. Reporting and interpretation of randomized controlled trials with statistically nonsignificant results for primary outcomes. JAMA 2010;303(20):2058-64.
- CrocettiMT, Amin DD, Scherer R. Assessment of risk of bias among pediatric randomized controlled trials. Pediatrics 2010;126(2):298-305
- Doria-Rose VP, Marcus PM, Miller AB, Bergstralh EJ, Mandel JS, Tockman MS, et al. Does the source of death information affect cancer screening efficacy results? A study of the use of mortality review versus death certificates in four randomized trials. Clinical Trials 2010;7(1):69-77
- Dwan K, Gamble C, Kolamunnage-Dona R, Mohammed S, Powell C, Williamson PR. Assessing the potential for outcome reporting bias in a review: a tutorial. Trials 2010;11:52
- Faggion CM, Listl S, Tu YK. Assessment of endpoints in studies on peri-implantitis treatment -- a systematic review. Journal of Dentistry 2010;38(6):443-50.
- Hemingway H, Philipson P, Chen R, Fitzpatrick NK, Damant J, Shipley M, et al. Evaluating the quality of research into a single prognostic biomarker: a systematic review and meta-analysis of 83 studies of C-Reactive protein in stable coronary artery disease. PLoS Medicine 2010;7(6):e1000286.
- Kirkham JJ, Dwan KM, Altman DG, Gamble C, Dodd S, Smyth R, et al. The impact of outcome reporting bias in randomised controlled trials on a cohort of systematic reviews. BMJ 2010;340:c365, 2010
- McGauran N, Wieseler B, Kreis J, Schuler YB, Kolsch H, Kaiser T. Reporting bias in medical research - a narrative review. Trials 2010;11:37.
- Song F, Parekh S, Hooper L, Loke YK, Ryder J, Sutton AJ, et al. Dissemination and publication of research findings: an updated review of related biases. Health Technol Assess 2010;14(8):iii, ix-iii,193
- Blackwell SC, Thompson L, Refuerzo J. Full publication of clinical trials presented at a national maternal-fetal medicine meeting: is there a publication bias? Am J Perinatol 2009;26(9):679-82.
- Chalmers I, Glasziou P. Avoidable waste in the production and reporting of research evidence. Obstet Gynecol 2009;114(6):1341-5.
- Decullier E, Chan AW, Chapuis F. Inadequate dissemination of phase I trials: a retrospective cohort study. PLoS Med 2009;6(2):e1000034
- Esposito E, Cipriani A, Barbui C. Outcome reporting bias in clinical trials. Epidemiol Psichiatr Soc 2009;18(1):17-8.
- Mathieu S, Boutron I, Moher D, Altman DG, Ravaud P. Comparison of registered and published primary outcomes in randomized controlled trials. JAMA 2009;302(9):977-84.
- Ohorodnyk P, Eisenhauer EA, Booth CM. Clinical benefit in oncology trials: is this a patient-centred or tumour-centred end-point? Eur J Cancer 2009;45(13):2249-52.
- Rasmussen N, Lee K, Bero L. Association of trial registration with the results and conclusions of published trials of new oncology drugs. Trials 2009;10:116.
- Ross JS, Mulvey GK, Hines EM, Nissen SE, Krumholz HM. Trial publication after registration in ClinicalTrials.Gov: a cross-sectional analysis. PLoS Medicine 2009;6(9):e1000144.
- Saad ED, Katz A. Progression-free survival and time to progression as primary end points in advanced breast cancer: often used, sometimes loosely defined. Ann Oncol 2009;20(3):460-4.
- Sun X, Briel M, Busse JW, Akl EA, You JJ, Mejza F, et al. Subgroup Analysis of Trials Is Rarely Easy (SATIRE): a study protocol for a systematic review to characterize the analysis, reporting, and claim of subgroup effects in randomized trials. Trials 200
- Taori G, Ho KM, George C, Bellomo R, Webb SAR, Hart GK, et al. Landmark survival as an end-point for trials in critically ill patients - comparison of alternative durations of follow-up: an exploratory analysis. Crit Care 2009;13(4):R128.
- Tschiesner UM, Rogers SN, Harreus U, Berghaus A, Cieza A. Comparison of outcome measures in head and neck cancer-literature review 2000-2006. Head and Neck 2009;31(2):251-9.
- Vedula SS, Bero L, Scherer RW, Dickersin K. Outcome reporting in industry-sponsored trials of gabapentin for off-label use. N Engl J Med 2009;361(20):1963-71
- Al-Marzouki S, Roberts I, Evans S, Marshall T. Selective reporting in clinical trials: analysis of trial protocols accepted by The Lancet. Lancet 2008;372(9634):201.
- Chan AW, Hrobjartsson A, Jorgensen KJ, Gotzsche PC, Altman DG. Discrepancies in sample size calculations and data analyses reported in randomised trials: comparison of publications with protocols. BMJ 2008;337:a2299.
- Chan AW. Bias, spin, and misreporting: time for full access to trial protocols and results. PLoS Med 2008;5(11):e230.
- Christensen E. Choosing the best endpoint. J Hepatol 2008;49(4):672-3.
- Corrales LA, Morshed S, Bhandari M, Miclau T. Variability in the assessment of fracture-healing in orthopaedic trauma studies. Journal of Bone and Joint Surgery American Volume 2008;90(9):1862-8.
- Dwan K, Gamble C, Williamson PR, Altman DG. Reporting of clinical trials: a review of research funders' guidelines. Trials 2008;9:66.
- Dwan K, Altman DG, Arnaiz JA, Bloom J, Chan AW, Cronin E, et al. Systematic review of the empirical evidence of study publication bias and outcome reporting bias. PLoS One 2008;3(8):e3081.
- Jureidini JN, McHenry LB, Mansfield PR. Clinical trials and drug promotion: selective reporting of study 329. International Journal of Risk and Safety in Medicine 2008;20(1-2):73-81.
- Llovet JM, Bisceglie AM, Bruix J, Kramer BS, Lencioni R, Zhu AX, et al. Design and endpoints of clinical trials in hepatocellular carcinoma. J Natl Cancer Inst 2008;100(10):698-711.
- Moineddin R, Butt DA, Tomlinson G, Beyene J. Identifying subpopulations for subgroup analysis in a longitudinal clinical trial. Contemporary Clinical Trials 2008;29(6):817-22.
- Peters J, Mengersen K. Selective reporting of adjusted estimates in observational epidemiology studies: reasons and implications for meta-analyses. Evaluation and the Health Professions 2008;31(4):370-89.
- Rising K, Bacchetti P, Bero L. Reporting bias in drug trials submitted to the Food and Drug Administration: review of publication and presentation. PLoS Med 2008;5(11):e217.
- Sinha I, Jones L, Smyth RL, Williamson PR. A systematic review of studies that aim to determine which outcomes to measure in clinical trials in children. PLoS Medicine 2008;5(4):e96.
- Tricco AC, Tetzlaff J, Sampson M, Fergusson D, Cogo E, Horsley T, et al. Few systematic reviews exist documenting the extent of bias: a systematic review. J Clin Epidemiol 2008;61(5):422-34.
- Bourgeois FT, Murthy S, Mandl KD. Outcome reporting among drug trials registered in ClinicalTrials.gov. Ann Intern Med 2010;153(3):158-66.
- Boutron I, Dutton S, Ravaud P, Altman DG. Reporting and interpretation of randomized controlled trials with statistically nonsignificant results for primary outcomes. JAMA 2010;303(20):2058-64.
- CrocettiMT, Amin DD, Scherer R. Assessment of risk of bias among pediatric randomized controlled trials. Pediatrics 2010;126(2):298-305
- Doria-Rose VP, Marcus PM, Miller AB, Bergstralh EJ, Mandel JS, Tockman MS, et al. Does the source of death information affect cancer screening efficacy results? A study of the use of mortality review versus death certificates in four randomized trials. Clinical Trials 2010;7(1):69-77.
- Dwan K, Gamble C, Kolamunnage-Dona R, Mohammed S, Powell C, Williamson PR. Assessing the potential for outcome reporting bias in a review: a tutorial. Trials 2010;11:52
- Faggion CM, Listl S, Tu YK. Assessment of endpoints in studies on peri-implantitis treatment -- a systematic review. Journal of Dentistry 2010;38(6):443-50.
- Hemingway H, Philipson P, Chen R, Fitzpatrick NK, Damant J, Shipley M, et al. Evaluating the quality of research into a single prognostic biomarker: a systematic review and meta-analysis of 83 studies of C-Reactive protein in stable coronary artery disease. PLoS Medicine 2010;7(6):e1000286.
- Kirkham JJ, Dwan KM, Altman DG, Gamble C, Dodd S, Smyth R, et al. The impact of outcome reporting bias in randomised controlled trials on a cohort of systematic reviews. BMJ 2010;340:c365, 2010
- McGauran N, Wieseler B, Kreis J, Schuler YB, Kolsch H, Kaiser T. Reporting bias in medical research - a narrative review. Trials 2010;11:37.
- Song F, Parekh S, Hooper L, Loke YK, Ryder J, Sutton AJ, et al. Dissemination and publication of research findings: an updated review of related biases. Health Technol Assess 2010;14(8):iii, ix-iii,193
- Blackwell SC, Thompson L, Refuerzo J. Full publication of clinical trials presented at a national maternal-fetal medicine meeting: is there a publication bias? Am J Perinatol 2009;26(9):679-82.
- Chalmers I, Glasziou P. Avoidable waste in the production and reporting of research evidence. Obstet Gynecol 2009;114(6):1341-5.
- Decullier E, Chan AW, Chapuis F. Inadequate dissemination of phase I trials: a retrospective cohort study. PLoS Med 2009;6(2):e1000034
- Esposito E, Cipriani A, Barbui C. Outcome reporting bias in clinical trials. Epidemiol Psichiatr Soc 2009;18(1):17-8.
- Mathieu S, Boutron I, Moher D, Altman DG, Ravaud P. Comparison of registered and published primary outcomes in randomized controlled trials. JAMA 2009;302(9):977-84.
- Ohorodnyk P, Eisenhauer EA, Booth CM. Clinical benefit in oncology trials: is this a patient-centred or tumour-centred end-point? Eur J Cancer 2009;45(13):2249-52.
- Rasmussen N, Lee K, Bero L. Association of trial registration with the results and conclusions of published trials of new oncology drugs. Trials 2009;10:116.
- Ross JS, Mulvey GK, Hines EM, Nissen SE, Krumholz HM. Trial publication after registration in ClinicalTrials.Gov: a cross-sectional analysis. PLoS Medicine 2009;6(9):e1000144.
- Saad ED, Katz A. Progression-free survival and time to progression as primary end points in advanced breast cancer: often used, sometimes loosely defined. Ann Oncol 2009;20(3):460-4.
- Sun X, Briel M, Busse JW, Akl EA, You JJ, Mejza F, et al. Subgroup Analysis of Trials Is Rarely Easy (SATIRE): a study protocol for a systematic review to characterize the analysis, reporting, and claim of subgroup effects in randomized trials. Trials 200
- Taori G, Ho KM, George C, Bellomo R, Webb SAR, Hart GK, et al. Landmark survival as an end-point for trials in critically ill patients - comparison of alternative durations of follow-up: an exploratory analysis. Crit Care 2009;13(4):R128.
- Tschiesner UM, Rogers SN, Harreus U, Berghaus A, Cieza A. Comparison of outcome measures in head and neck cancer-literature review 2000-2006. Head and Neck 2009;31(2):251-9.
- Vedula SS, Bero L, Scherer RW, Dickersin K. Outcome reporting in industry-sponsored trials of gabapentin for off-label use. N Engl J Med 2009;361(20):1963-71
- Al-Marzouki S, Roberts I, Evans S, Marshall T. Selective reporting in clinical trials: analysis of trial protocols accepted by The Lancet. Lancet 2008;372(9634):201.
- Chan AW, Hrobjartsson A, Jorgensen KJ, Gotzsche PC, Altman DG. Discrepancies in sample size calculations and data analyses reported in randomised trials: comparison of publications with protocols. BMJ 2008;337:a2299.
- Chan AW. Bias, spin, and misreporting: time for full access to trial protocols and results. PLoS Med 2008;5(11):e230.
- Chan AW, Hrobjartsson A, Haahr MT, Gotzsche PC, Altman DG. Empirical evidence for selective reporting of outcomes in randomized trials: comparison of protocols to published articles. JAMA 2004; 291(20):2457-65.
- Chan AW, Krleza-Jeri K, Schmid I, Altman DG. Outcome reporting bias in randomized trials funded by the Canadian Institutes of Health Research. CMAJ 2004; 171(7): 735-40.
- Chan AW, Altman DG. Identifying outcome reporting bias in randomised trials on PubMed: review of publications and survey of authors. BMJ 2005; 330(7494): 753.
- Christensen E. Choosing the best endpoint. J Hepatol 2008;49(4):672-3.
- Corrales LA, Morshed S, Bhandari M, Miclau T. Variability in the assessment of fracture-healing in orthopaedic trauma studies. Journal of Bone and Joint Surgery American Volume 2008;90(9):1862-8.
- Dwan K, Gamble C, Williamson PR, Altman DG. Reporting of clinical trials: a review of research funders' guidelines. Trials 2008;9:66.
- Dwan K, Altman DG, Arnaiz JA, Bloom J, Chan AW, Cronin E, et al. Systematic review of the empirical evidence of study publication bias and outcome reporting bias. PLoS One 2008;3(8):e3081.
- Jureidini JN, McHenry LB, Mansfield PR. Clinical trials and drug promotion: selective reporting of study 329. International Journal of Risk and Safety in Medicine 2008;20(1-2):73-81.
- Llovet JM, Bisceglie AM, Bruix J, Kramer BS, Lencioni R, Zhu AX, et al. Design and endpoints of clinical trials in hepatocellular carcinoma. J Natl Cancer Inst 2008;100(10):698-711.
- Moineddin R, Butt DA, Tomlinson G, Beyene J. Identifying subpopulations for subgroup analysis in a longitudinal clinical trial. Contemporary Clinical Trials 2008;29(6):817-22.
- Peters J, Mengersen K. Selective reporting of adjusted estimates in observational epidemiology studies: reasons and implications for meta-analyses. Evaluation and the Health Professions 2008;31(4):370-89.
- Rising K, Bacchetti P, Bero L. Reporting bias in drug trials submitted to the Food and Drug Administration: review of publication and presentation. PLoS Med 2008;5(11):e217.
- Sinha I, Jones L, Smyth RL, Williamson PR. A systematic review of studies that aim to determine which outcomes to measure in clinical trials in children. PLoS Medicine 2008;5(4):e96.
- Tricco AC, Tetzlaff J, Sampson M, Fergusson D, Cogo E, Horsley T, et al. Few systematic reviews exist documenting the extent of bias: a systematic review. J Clin Epidemiol 2008;61(5):422-34.
- Furukawa TA, Watanabe N, Omori IM, Montori VM, Guyatt GH. Association between unreported outcomes and effect size estimates in Cochrane meta-analyses. JAMA 2007; 297(5): 468-70.
- Gøtzsche PC. Multiple publication in reports of drug trials. Eur J Clin Pharmacol 1989; 36:429-32.
- Hahn S, Williamson PR, Hutton JL, Garner P, Flynn EV. Assessing the potential for bias in meta-analysis due to selective reporting of subgroup analyses within studies. Stat Med 2000; 19(24): 3325-36.
- Hahn S, Williamson PR, Hutton JL. Investigation of within-study selective reporting in clinical research: follow-up of applications submitted to a local research ethics committee. Journal of Evaluation in Clinical Practice 2002; 8(3): 353-9.
- Hrobjartsson A, Chan AW, Haahr MT, Gotzsche PC, Altman DG. [Selective reporting of positive outcomes in randomised trials--secondary publication.. A comparison of protocols with published reports]. [Danish]. Ugeskr Laeger 2005; 167(34): 3189-91.
- Hutton J and Williamson P. Bias in meta-analysis due to outcome variable selection within studies. Applied Statistics 2000; 49: 359-370.
- Williamson PR, Gamble C. Identification and impact of outcome selection bias in meta-analysis. Stat Med 2005; 24(10): 1547-61.
Quality of Reporting and Reporting Guidelines
- Altman DG. Poor-quality medical research: what can journals do? JAMA 2002; 287(21): 2765-7.
- Balk EM, Bonis PA, Moskowitz H, Schmid CH, Ioannidis JP, Wang C, et al. Correlation of quality measures with estimates of treatment effect in meta-analyses of randomized controlled trials. JAMA 2002; 287(22): 2973-82.
- Gluud C, Nikolova D. Quality assessment of reports on clinical trials in Journal of Hepatology. J Hepatol 1998;29:321-327.
- Gluud C, Kjaergard LL. Quality of trials in portal hypertension and other fields of hepatology. 3rd Baveno International Consensus Workshop. Portal Hypertension into the Third Millennium. Definition, Methodology and Therapeutic Strategies in Portal Hypertension. Blackwell Science: Oxford. 2001;204-218.
- Gluud LL, Sørensen TI, Gøtzsche PC, Gluud C. The journal impact factor as a predictor of trial quality and outcomes: cohort study of hepatobiliary randomized clinical trials. Am J Gastroenterol 2005;100(11):2431-5.
- Gluud C. The culture of designing hepato-biliary randomised trials. J Hepatol 2006;44(3):607-15.
- Gluud C, Klingenberg SL, Gluud LL. Quality of randomised clinical trials in portal hypertension and other fields of hepatology. In: Roberto De Franchis, ed. Portal Hypertension IV. 1st ed. Oxford: Blackwell Publishing Ltd; 2006;328-344.
- Gluud LL. Bias in clinical intervention research. Am J Epidemiol 2006;163:493-501.
- Huwiler-Muntener K, Juni P, Junker C, Egger M. Quality of reporting of randomized trials as a measure of methodologic quality. JAMA 2002; 287(21): 2801-4.
- Jüni P, Altman DG, Egger M. Systematic reviews in health care: Assessing the quality of controlled clinical trials. BMJ 2001; 323(7303): 42-6.
- Jüni P, Witschi A, Bloch R, Egger M. The hazards of scoring the quality of clinical trials for meta-analysis. JAMA 1999; 282(11): 1054-60.
- Kjærgard LL, Nikolova D, Gluud C. Randomized clinical trials in Hepatology ? predictors of quality. Hepatology 1999;30:1134-38.
- Kjaergard LL, Villumsen J, Gluud C. Reported methodologic quality and discrepancies between large and small randomized trials in meta-analyses. Ann Intern Med 2001; 135(11): 982-9.
- Kjaergard LL, Frederiksen S, Gluud C. Validity of randomized clinical trials in Gastroenterology from 1964-2000. Gastroenterology 2002;122:1157-1160.
- Liu XM, Li YP, Yu XT, Feng J,Zhong XS,Yang SY, Li J. Assessment of registration quality of trials sponsored by China. Journal of Evidence-Based Medicine, 2009; 2(2):8-18.
- Moher D, Pham B, Jones A, Cook DJ, Jadad AR, Moher M, et al. Does quality of reports of randomised trials affect estimates of intervention efficacy reported in meta-analyses? Lancet 1998; 352(9128): 609-13.
- Siersma V, Als-Nielsen B, Chen W, Hilden J, Gluud LL, Gluud C. Multivariable modeling for meta-epidemiological assessment of the association between trial quality and treatment effects estimated in randomised clinical trials. Statistics in Medicine 2007; 26(14): 2745-58.
For more information refer to the EQUATOR network for resources for good reporting.




