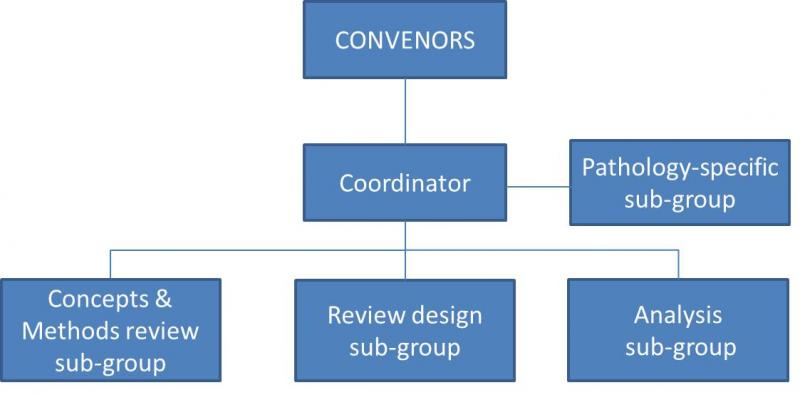
The Concepts and Methods Review Sub-Group will:
- Produce a glossary of HRQL terms, that could be included in the current Cochrane glossary
Chair: E. Mc Coll
Members: G. Bertolotti, F. Efficace, L. Maxwell, G. Paterson, O. Patiño, R. Oates-Whitehead, P. Tugwell, A. Wenban, A. Lloyd, N. Chaiyakunapruk
The Review Design Sub-Group will:
- Collect examples of clinical trials that incorporate poor and good quality PRO assessment
- Develop and assess a checklist to evaluate the quality of PRO outcomes included in RCTs and retrieved by Cochrane reviewers. This checklist will provide criteria that enable reviewers to include or exclude PRO studies. This work may be performed in close collaboration with members of the European Regulatory Issues and Quality of Life Assessment (ERIQA) Project, the mission statement of which is « establishing principles and practices for the integration of Health-related Quality of Life outcomes in the regulatory process » (see ERIQA Group, and ISOQOL and ISPOR.)
- Develop guidelines on psychometric criteria, appropriateness and cross-cultural validation of instruments for addressing PRO issues in Cochrane reviews
- Develop and assess checklists for quality assessment of systematic reviews including PRO outcomes
Chair: X. Badia
Members: R. Acosta, G. Bertolotti, A. Bottomley, P. Fayers, Z. Fedorowicz, S. Green, G. Guyatt, A. Hareendran, S. Hunt, A. Ismail, L. Maxwell, V. Kennedy, E. McColl, P. Marquis, M. Pandey, O. Patiño, M. Sullivan, P. Tugwell
The Analysis Sub-Group will:
- Develop and validate methods for pooling PRO data in the context of a meta-analysis
- Develop guidelines for the interpretation of PRO results [i.e. Effect Size (ES), Minimally Important Difference (MID),Number of Patients Needed to Treat (NNT) ]
- Develop methods for the inclusion of data into RevMan software (non-numeric data, as for MID, SMD, for each domain or with a summary score)
Chair:Chair: J.Sloan
Members: D. Beaton, O. Chassany, Desmond Curran, P. Fayers, G. Guyatt,, D. Patrick, M. Rothman, M. Sullivan, A. Hakuzimana, N. Chaiyakunapruk
The Pathology-specific Sub-Groups will:
- Help reviewers to develop protocols and reviews where it has been decided to include PRO outcomes.
Members:
CNS: A. Hareendran, M. Sullivan
Diabetes: E. McColl, M. Sullivan
Elderly: A. Hareendran, D. Patrick, E. McColl, M. Sullivan
Gastro-intestinal: O. Chassany, P. Marquis
HIV: X. Badia, S. Hunt
Musculoskeletal: D. Beaton, S. Green
Oncology/Palliative Care: K. Anie, A. Bottomley, P. Fayers, Manoj Pandey, R. Oates-Whitehead
Ophthalmology: R. Acosta
Psychiatry: M. Rothman
Respiratory Diseases: S. A. Antoniu, G. Bertolotti, M. Carone, O. Chassany, C. Donner, M. Sullivan
Urology: P. Marquis, D. Patrick
